Toxoplasma gondii (Toxoplasmosis)
Authors: Helieh S. Oz, DVM, PhD, AGAF
Authors, Second Edition: Jose G. Montoya, M.D., Jacques Couvreur, M.D., Catherine Leport, M.D.
ETIOLOGY
Toxoplasma gondii is an intracellular organism which infects warm blooded animals and humans. Toxoplasma is one of the most important sources of foodborne diseases and congenital infection. An estimated 1.5 billion people globally are infected, frequently with unknown lifelong health complications (86, 150). Toxoplasma is “Category B pathogenic agent” according the Center for Disease Control (CDC), and National Institute of Health (NIH). Following acute phase, the organisms dwell mainly in muscles and brain in cyst forms for the lifetime awaiting to be reactivated. Toxoplasma was discovered a century ago (1908) by Nicolle and Manceaux in an African rodent. Despite ever since ongoing investigations, yet there is no safe and effective therapy for congenital, chronic infection or a vaccine available to prevent toxoplasmosis.
Toxoplasma is an Apicomplexan protozoan parasite with sexual stage which takes place after oral infection in the intestinal epithelia of cats, as organisms replicate and mature to form resistant oocysts passed in the feces (68). Oocysts are formed by gametogony and sporogony. Cats usually become infected within 6 months of their life and pass unsporulated (immature) oocysts for about 1-2 weeks. Approximately millions of oocysts are excreted in the feces of cat 5 to 20 days after initial infection. Unsporulated oocysts become sporulated and mature in about 1 to 2 days in the moist soil to become infective forms of sporozoites. Sporozoites within protective oocyst are highly infectious when ingested and oocysts can remain viable in warm moist soil for over 1 year.
Humans and animals acquire systemic form of infection in asexual stage through consumption of contaminated animal products including fresh untreated milk, raw/undercooked meat and seafood or mature oocysts in water, fruits and vegetables (52, 54, 150). After sporozoites are released in the body they transform into tachyzoites, and attack every type of nucleated cells. Tachyzoites are the proliferative form in acute stage and reactivated stage of chronic infection. Bradyzoites are shaped in the dormant cysts during inactive or chronic stage of infection which replicate more slowly than the tachyzoites. Small pseudocysts are formed in tissues mainly during the early infection about a week after infection. Tissue cysts are formed within the host cells after acute stage and may contain thousands of bradyzoites which can persist as viable organisms as long as the host lives (152, 172).
Presence of the free tachyzoites establishes an active toxoplasmosis susceptible to available anti-Toxoplasma drugs. Tissue cysts are formed within the host cells which protect the bradyzoites against antimicrobial agent. The slow replicating bradyzoites are protected by the cyst wall against the action of the most available antimicrobial drugs. Bradyzoites are less susceptible to destruction by proteolytic enzymes than tachyzoites (52). Tissue cysts detection in biopsy specimens in addition to presence of inflammatory cells in the vicinity of the cysts determine patient with reactivation of dormantToxoplasma infection. Sporozoites are highly infectious when ingested. Oocysts can remain viable in environment for long period of time.
Toxoplasmosis Transmission
Toxoplasmosis is not acquired by casual direct contact from infected person with intact skin or airborne.
The common routes of transmission include:
a. Foodborne by contaminated water, milk, meat, unwashed fruits and vegetables or utensils.
b. Fetal Maternal (congenital).
c. Cat-man and animals: By handling contaminated soil, water and accidental fecal-oral infection.
d. Contaminated blood and organ transplant transmission.
e. Less common routes are “Sexual Transmission” by contaminated semen, and body fluid.
f. Accidental lab acquired infection by inoculation of contaminated material.
g. Breast feed transmission through contaminated milk with cysts and tachyzoites or environmental contamination of udders with oocysts.
Toxoplasma tachyzoites are secreted into milk in various farm animals. Contaminated milk transmission from farm animals has been reported by the consumption of unpasteurized milk from livestock as well as less common farm animals like camels and donkeys’ milk (41, 53, 187). Tachyzoites survive in the fresh milk and milk products in refrigeration for several days and can transmit the infection. Appropriate heating, freezing, or pasteurization can eliminate the infective forms for safe consumption.
EPIDEMIOLOGY
Toxoplasmosis is a global disease which burden humans and animals. The infection is usually asymptomatic and mild, while the prevalence of exposure increases with age and the lifestyle. There are conflicting reports regarding the decline of the seroprevalence rates of Toxoplasma infection in United States. It is estimated 22.5% of the U.S.A. population 12 years and older have been infected withToxoplasma (31). However, the infection report is on the rise in Europe (50% to 75%), Africa, South America, and Asia where the rate of infection is as high as 90% (181) possibly due to better diagnostic techniques and awareness. Toxoplasma infection is endemic in the farms and rural areas representing poverty and lack of appropriate hygiene. In the United States, the age-adjusted Toxoplasmaseroprevalence among people from 6 to 49 years old was reported to be 10.8% (95% confidence limits 9.6%, 11.9%), and for child-baring women 15 to 44 years old 11.0% (95% confident limit 9.5%, 12.4%) (101). Amongst immunocompetent individuals, the initial infection usually leads to a latent chronic infection that is effectively controlled by the host immune system. While, babies and immunosuppressed individuals are in a higher risk for active toxoplasmosis. The immunosuppressed ones include transplant recipients (bone marrow and solid organ transplants), patients with hematologic malignancy (e.g. Hodgkin's disease), and AIDS patients (70). The incidence of clinical manifestation of toxoplasmosis is uncommon among patients with CD4+ counts >200 cells/µl. Toxoplasmaencephalitis has been reported in 30% of AIDS patients with CD4+ counts of less than 100 cells/µl and who are not receiving effective anti-Toxoplasma prophylaxis or highly active antiretroviral therapy (HAART) (73). The incidence of Toxoplasma-induced encephalitis and the rate of mortality have significantly declined in United States and Europe where HAART and prophylactics for opportunistic infections including Toxoplasma are used and the guidelines for the use are appropriately implemented.
Humans and animals become infected by ingestion of contaminated food or hands with oocysts excreted from infected cats or consumption of undercooked meat containing cysts. Sporulated oocysts are transmitted to humans through soil during gardening, handling infected cat’s feces, contaminated fruits and vegetables or unpurified water (12, 20). Indeed, contaminated water is reported as a major source for infection during pregnancy in rural area (3). In the United States most farm animals including pig, lamb, goat, chicken and game animals were found infected to harbor Toxoplasma cysts (41, 52).
CLINICAL MANIFESTATIONS
Immunocompetent Patients
Acute toxoplasmosis is mostly subclinical and unnoticed in immune-intact humans and animals with flue like symptoms, fever, and cervical lymphadenopathy. The symptoms may last for a few weeks or up to months. In some individuals the clinical symptoms can be broad and mimic viral diseases such as infectious mononucleosis, cytomegalovirus and human herpes virus-6 with manifestation of sore throat, splenohepatomegaly, and presence of immature lymphocytes in blood stream. In addition, patients may develop symptoms of hepatitis, pancreatitis, splenitis, myocarditis, myositis and pain (31, 148,150, 151). Toxoplasmosis can be suspected in those patients with immunosuppressive conditions or unexplained syndromes in those who travelled to unsanitary and rural areas.
Maternal and Fetal Toxoplasmosis
Pregnant women become infected by ingesting oocysts containing sporozoites contaminated food and water or consuming cysts with bradyzoites infected undercooked meat. The acute toxoplasmosis can be subclinical and diagnosed by routine serologic examinations. The primary infection acquired during pregnancy can be transmitted and infect the fetus via the placenta. However, normal women (immunocompetent) who were exposed to infection in past and prior pregnancy with low or stable anti-Toxoplasma IgG antibody titers are immune and mostly deliver healthy neonates.
Additionally, transplacental transmission occurs by the reactivation of the chronic or latent infection caused by stressors due to pregnancy and other immunsuppressors. In any cases, whether oocysts or cysts are the origin of the infection, the organisms transform to active stage of tachyzoites and propagate to reach uterus. Tachyzoites then penetrate the transplacental blood barrier and invade the fetus to compromise the embryonic development. Toxoplasma infection rate of child-bearing women in Brazil is estimated 50% to 80%, and about 50% of children have antibodies titers. Also, 5 to 23 infants are found to be infected per 10,000 at birth in Brazil (54). Contaminated food and water with Toxoplasma oocysts are reported as the main source of human infection in North America (22, 130). The National Collaborative Chicago-based Congenital Toxoplasmosis (NCCCTS) reported that 78% of moms with infected infants had primary infection originated from oocysts while only half of these moms had direct contact with cats America (22, 130). While, educational hygienic programs are beneficial to the community, by itself cannot protect health and socioeconomic consequences related to the toxoplasmosis. Therefore, an effective and practical global cat vaccination program, implementation of serological testing of pregnant women, newborns, and those in high risk of infection followed by effective preventive and therapeutic regimens are necessary to eradicate toxoplasmosis.
Congenital Toxoplasmosis
Congenital disease was first reported in a 3 days old baby girl in New York, 1938 who developed seizure and eyes lesion and died after one month of birth (214). Congenital toxoplasmosis is mostly a subclinical infection in about 85% of the infants whose mothers have been treated for Toxoplasma during pregnancy (143). Theses proportions are influenced by factors such as, the time of maternal infection, treatment given to the mother, organism load and the genetics of the host and the strain ofToxoplasma. The incidence of fetal infection is estimated 2% if the maternal infection occur before week 10 and 90% in the last weeks of pregnancy. However, early maternal infection is reported to cause higher risk of disease and brain damage. Congenital toxoplasmosis manifests with spontaneous miscarriage and abortion, fetal growth retardation, encephalomyelitis, intracranial calcifications, hydrocephalus, neurological, mental illnesses, retinochoroiditis, visual and auditory inflammatory disorders, cardiovascular abnormalities (55, 130, 150, 152, 174), gastrointestinal complications and pain (57, 114, 148, 149, 150, 158). The severity of complications depends on the gestation period when the fetus is infected, and the earlier the infection the more severe the complications (55, 174). Those fetuses infected late in gestation may born normal, but develop symptoms of CNS and retinochoroiditis later in life and the new lesions may occur in untreated as well as treated children (55). Retrospective trials (2000–2011) reviled 18% (2206/12035) prevalence rate of anti-Toxoplasma antibody in pregnant women. Thirty eight per 10,000 of these women had acute infection and 5.8% transplacentally infected their neonates as reported in Argentina (29).
Other factors involve are genetic tendency affecting immune response including HLA may influence clinical outcomes (99, 123). As pregnancy progresses, maternal immune system is modified to tolerate the growing semi-allogeneic fetus, at the same time to protect mom and offspring against pathogens. Therefore, pregnancy requires a complex balance in regulation of the immune system at the fetal maternal utero-embryonic border ensuring tolerance (Th2) of the fetus (142, 150). The HLA-G expression reprograms maternal immune response towards tolerance. Professional antigen presenting B cells can activate or silent T cells, to partake in regulation of the immune response. B cells are involved in humoral immune response leading to pre-eclampsia, during pregnancy (2).
Spiramycin therapy can prevent maternal transmission of infection in infants, particularly when administered within 8 weeks of seroconversion (199). The proportion of infected newborns was 61% from untreated-infected mothers and 23% in the group receiving spiramycin treatment (36). Retinochoroiditis clinical cases can occur in up to 80% of the infants born with congenital toxoplasmosis cases with subclinical symptoms, particularly during puberty and adolescence (135, 213), which can be prevented by early diagnosis and treatment of the children (80, 207). Isolation of Toxoplasma from the placenta is associated with Toxoplasma infected infant. The proportion of infected babies depends on the early diagnosis and treatment given during pregnancy. Usually 90% of untreated-infected moms deliver infected babies, in contrast 25% of treated with spiramycin and 50% of those moms receiving spiramycin and the combined pyrimethamine plus sulfadiazine in alternating courses deliver seronegative infants (36).
Ocular Manifestation of Disease
Retinochoroiditis often occurs in congenital infection in neonates but it can also happen post parturition as acquired toxoplasmosis in immunocompetent children or adults (18, 61, 137). About 21% of adults in Southern Brazil were found infected. In immunosuppressed and immunodeficient individuals particularly AIDS patients ocular involvement may occur due to relapse of a previously-acquired infection. Patients, often develop multiple active, atypical, large atypical and bilateral retinal lesions (145). Many patients may develop ocular disease soon after Toxoplasma infection. From 205 patients (10-96 years old) tested for ocular Toxoplasmosis between 2004-2010 at Palo Alto Medical Foundation Toxoplasma Reference Laboratory, 11.7% were detected to have recent infection (100).
Before the AIDS pandemic only 50 cases of toxoplasmosis were documented in immunosuppressed patients (121, 182). Then the number of diagnosed cases significantly increased to several thousands of cases afterward. Clinical presentation of toxoplasmosis in these patients varies, as a prolonged unexplained fever to visceral manifestations, with neurological and pulmonary lesions (126).
In HIV and AIDS patients, the major findings are Toxoplasma-induced encephalitis, as well as ocular, pulmonary or disseminated disease (118, 119). In a French hospital database on HIV with 60,000 patients, the incidence changed from 29% in 1992 to 14% in 1995 and 2.6% in 1998. This decline in the number of patients with toxoplasmosis was due to the advancement of HAART in 1995 as well as other prophylactic compounds such as combined trimethoprim plus sulfamethoxazole. Patients with encephalitis may develop symptoms of headache, fever, and neurological symptoms, including dizziness, confusion, coma, motor defect, visual abnormalities, and seizures.
Immunosuppressed Patients
Patients with dormant toxoplasmosis who receive allogeneic hematopoietic stem cell transplants or those with graft versus host disease are at risk of severe or fatal toxoplasmosis by reactivation of their latent infection. Toxoplasmosis can be manifested with fever, pneumonia or brain cysts which should alert the healthcare staff of infection. Seronegative patients who received a seropositive donor organ are similarly at risk of toxoplasmosis.
Organ Transplantation, Blood Transfusion and Toxoplasmosis
Organ transplant patients with liver, lung, bone marrow, pancreas and kidney recipients are at risk for toxoplasmosis. This is as a result of immunosuppressive treatments and organ from infected donors or reactivated chronic infection followed with a high morbidity and mortality rate if not detected in early stages. Toxoplasmosis is considered as underestimated complication following hematopoietic stem cell transplantation and blood transfusion (195). It is commonly detected in autopsy or remains undetected due to the general and non-specific symptoms combined with lack of clinical awareness of healthcare personals (140). In Japan, while the incidence rate of toxoplasmosis in transplant patients is low (1.8%) but the mortality in infected patients reported to be very high (195). Toxoplasmosis is frequently difficult to diagnose and potentially fatal in kidney transplant recipients.
Women with renal disease who become pregnant are generally in a very high risk especially in those with chronic renal insufficiency, end-stage renal disease, dialysis patients and transplant recipients. Hypertension is the most common life-threatening problem in these moms. Transplant recipients are at risk for infections including Toxoplasma and can infect their fetus.
Renal disease patients have an increased risk for prematurity and intrauterine growth restriction. For women who conceive after starting dialysis, the probability of a surviving infant is reduced to 50% (89,90). Therefore, transplant recipients including allogeneic bone marrow and peripheral blood stem cell for malignant hematologic diseases and severe congenital immunodeficiency conditions need to be monitored for Toxoplasma as well as other opportunistic infections (90).
Mental Diseases and Toxoplasmosis
Toxoplasma attacks the central nervous system with adverse affect in brain neuro-structural development and pathological, as well as psycho-behavioral alteration, leading to mental retardation (11, 24, 211). More recently, maternal exposure or latentToxoplasma infection has been linked to a potential increased risk for autism and schizophrenic episodes and this hypothesis has received a great deal of scientific and media coverage (62). Toxoplasma can alter brain dopamine (59, 203). In a longitudinal trial, women with chronic toxoplasmosis had a high risk of self-harm and accidents (162). Another cross-sectional study indicated that Toxoplasma seropositive individuals had elevated risk of nonfatal suicidal violence (216).
Women with chronic toxoplasmosis have higher proportion to deliver infants with genetic or developmental disorders. For instance premature fetuses show slower rates of development in the first trimester as well as postnatal motor development in infants of moms with latent toxoplasmosis. In addition, these infants had a significant delayed in ability to control their head movements (p<0.05), due to decreased strength of embryonic quality control related to immunosuppression caused by infection in mothers. These defects may be due to severity of congenital toxoplasmosis, cognitive and developmental deficits or caused by the diarrheal malnutrition (103).
A 20 year retrospective trial with military personnel, university students, pregnant women and blood donors has shown even asymptomatic Toxoplasma infection can greatly affect humans (62). The infected subjects varied from controls in the characteristic behaviors suggesting Toxoplasma influences personality and increasing probability of traffic accidents of infected individuals. Dormant infection in mothers is associated with immunosuppression, and high prevalence to have children with Down syndrome or schizophrenia (62).
Another trail evaluated school children in rural area for seropositive and seronegative, developmental age, body mass, height, physical fitness, grades and the risk factors of infection like contact with house cats and consumption of undercooked meat products. The prevalence was 41% for children to have anti-Toxoplasma antibodies measured by IFAT and ELISA tests. The rate of infection was lower in boys (36%) than in girls (44%). The developmental age measures with electrophoretical mobility of nuclei and school performance was significantly lower for Toxoplasma infected girls compared to matched controls. Those boys who ate raw/undercooked meat products were most likely to be seropositive (134). Another observational study (2000-2012) from Nepal, declared congenital toxoplasmosis as second to malaria with highest parasitic burden of 9255 disability-adjusted life year of 95% (48).
Toxoplasmosis and Autoimmune Disease
While, etiological factors in the induction of autoimmune disease remain obscure a possible link with toxoplasmosis may exist. Immunosuppressants, and monoclonal antibodies such as anti-TNF are extensively used in the treatment of autoimmune diseases and organ transplantation may provoke acute toxoplasmosis in these patients. Sera of 1514 patients with 10 autoimmune diseases in Europe and Latin America and 437 controls investigated for the prevalence of auto-antibodies and anti-Toxoplasma antibodies IgG and IgM in serum (186). Anti-Toxoplasma antibody IgG was positive in 42% of patients versus 29% of controls (p<0.0001). Anti-Toxoplasma antibody IgM was more prevalent in patients with anti-phospholipid syndrome (p<0.01), systemic sclerosis (p<0.05) and inflammatory bowel disease (p<0.05), than in controls. Anti-Toxoplasma antibody IgG was associated with ANCA-associated vasculitides (p<0.01), anti-phospholipid syndrome (p<0.0001), autoimmune thyroid diseases (p<0.0001), systemic sclerosis (p<0.0001), and rheumatoid arthritis (p<0.0001). The findings support the notion that Toxoplasma may contribute to the pathogenesis of autoimmune disease (150, 152, 186). Chronic inflammatory bowel disease (IBD) is mainly Crohn’s disease and ulcerative colitis. Chronic inflammatory bowel disease is considered as an exaggerated immune response to gut microbiota, and toxins from gram negative lipopolysaccharid (LPS), bypassing the inflamed mucosa (23, 153, 154). Additionally, extensive use of immunosuppressive agents in IBD patients increases the possibility to develop opportunistic diseases and toxoplasmosis. The greatest risks of infections may relate to the combined use of immunomodulating agents rather than to individual drugs (57). Chronic inflammatory bowel disease patients treated with corticosteroids, azathioprine, methotrexate, cyclosporine, and TNF-blocking biological are susceptible to infections. Crohn’s patients are prone for gut abscess formation followed by infectious complications as well as toxoplasmosis. This alerts the physicians treating patients with IBD to be aware of the risk for infectious as well as of strategies to minimize these infectious complications in these patients (57, 150).
Severe colonic inflammatory response is observed in pregnancy model for toxoplasmosis, associated with significant shortening in colonic length, with infiltration of lymphocytes, and macrophages and microabscess formations in the cryptic structures in infected dams (148, 149, 157, 158). Chemically induction chronic inflammatory bowel disease model for ulcerative colitis utilizes oral administration of dextran sulfate sodium (DSS) for 3-16 cycles to induce chronic inflammation in the gut (153, 154) with increased gram negative gut microbiota. Colitis significantly increases colonic LPS (550-fold) in concentrations. Similarly, Toxoplasma induced ileitis with significant elevated ileal concentrations LPS (3,300-fold) P<0.01 (58). Sera from 119 chronic inflammatory bowel disease patients and 98 controls were assessed for anti-Toxoplasma antibody. Titer of anti-Toxoplasma was found higher in chronic inflammatory bowel disease patients than controls suggesting possible involvement ofToxoplasma in the pathogenesis of chronic inflammatory bowel disease, and specifically in Crohn’s disease (114). Finally, while inflammation is essential mechanism of defense against pathogens, excessive and chronic inflammatory response as in Toxoplasma infection severely damages the tissues and organs similar to as observed in patients with autoimmune diseases (30, 150). Toxoplasmainfection causes an exaggerated Th1 systemic inflammatory response which enhances pro-atherogenic effects (115, 170).
Toxoplasma and Sexual Attraction
Recent investigations reveal mind alteration and sexual attraction in Toxoplasma infected rats to seek cat’s urine odor, while normal rats fear and flight as innate survival defensive (91, 106, 150). Therefore, brain altered and fearless infected rodent is eaten by cats’ species (definitive host) to complete the organism’s sexual lifecycle. Toxoplasma impairs the limbic brain neurons responsible for instinct defensive behavior and judgment activity adjacent to limbic regions of sexual desire when exposed to cat’s odor (91).
Toxoplasmosis is sexually transmitted to the partner by contaminated semen during natural mating. Also, artificial insemination of semen from infects animals’ results in vertical transmission with 80% embryonic growth disruption and abortion (4, 117, 209). In fact, sexual transmission with infected semen during natural mating as well as artificial insemination remains as a potential route for vertical transmission to progeny in humans. Additionally, pregnant women with latent infection have a higher risk of infants with genetic or developmental disorders such as premature and slow motor development. These defects may be linked to malnutrition due to diarrhea and gut disorders or congenital toxoplasmosis induced developmental deficits (103, 152).
LABORATORY AND RADIOLOGICAL DIAGNOSTICS
Toxoplasma infection is a serious health risk issue for the fetus and immunocompromised patients. Therefore, rapid and accurate diagnostic measures are required for possible preventions and available therapeutic modalities. Toxoplasma infection and toxoplasmosis can be confirmed by the use of various methodologies including serological tests, polymerase chain reaction (PCR), and histological exams, as well as isolating the organisms and imaging analysis. Diagnosis of toxoplasmosis is mainly based on the serological tests detecting anti-Toxoplasma-specific antibodies in the patients’ sera samples. Most of the commercial serological kits currently available are based on Toxoplasma lysate antigens (137). Lab tests are less reliable in the immunosuppressed patients which diagnosis performed in the context of the clinical development.
Serologic Diagnosis for Toxoplasma Antibodies
The detection of Toxoplasma-specific IgM, IgG, IgA are to establish the infection in patients exposed to the organisms. Patients with negative IgG and IgM do not have serological evidence of prior exposure to the organism and are considered as uninfected; except for patients with organ transplant which serological tests alone are not reliable. Patients with positive IgG but negative IgM are possibly infected for several months or years. Positive or negative IgG and negative IgM serological tests results obtained in commercial laboratories are usually reliable (113, 171). Positive IgM test may indicate a recent acquired or an activated infection in the patients. In neonates a positive IgM test 5 days or a positive IgA after 10 days of birth confirms congenital toxoplasmosis.
Sabin-Feldman dye test, a complement-lysis-based assay with relative sensitivity and specificity to measure anti-Toxoplasma IgG antibodies is the international “gold standard” for diagnosis of toxoplasmosis (183). The Sabin-Feldman dye test is considered to be somehow more reliable than commercially available ELISA kits in some labs, but the test is time consuming and requires live organisms treated with each serum dilution to be analyzed under the microscope (38).
Available commercial kits require parasite antigen and have interassay variability. The recombinant Toxoplasma SAG1 antigen expressed in bacteria as inclusion bodies, and refolded in native form by dialysis has shown promising results. The antigen used in ELISA can detect IgG in saliva and sera with 100% sensitivity and specificity (32). In addition, the new multiplex, BioPlex® 2200 automated analyzer (Bio-Rad Laboratories, Hercules, CA) and ToRC IgG/IgM kit can detect anti-Toxoplasma antibodies in the same assay and is considered superior to Platelia IgG/IgM ELISA test (Bio-Rad Laboratories) or Toxo-Screen Direct Agglutination assay (bioMérieux, Lyon, France) with 91.3% specific and 97.8% sensitive to detect IgG and IgM, thus yielding more precise diagnosis of acute Toxoplasma infection (82).
Isolation of Toxoplasma
Detection of the organisms establishes an acute infection. Isolation can be done by tissue culture or mouse inoculation. Cell culture is more widely available, but mouse inoculation is more sensitive. Bradyzoites in cysts and free tachyzoites in infected tissue biopsies are detected using immunohistochemical staining, Giemsa and other direct staining methodologies.
Immunohistochemical Staining (IHC)
IHC is a reliable technique for possible visualization of organisms in biopsies or sections. The paraffin-embedded sections are cut, deparaffinized with xylene, rehydrated in alcohol baths, washed in PBS with 0.1% bovine serum albumin (BSA), then quenched for endogenous peroxidase activity by incubating in 3% hydrogen peroxide in methanol 30 min, and blocked with rabbit serum (Dako #N1699) for 30 min. The sections are incubated with poly or monoclonal anti-Toxoplasma antibody, diluted 1:500 for 90 min and developed with DAB-Chromogen (DakoCarpenteria, CA) for about 5 min until signal developed and subsequently counterstained with hematoxylin then ammonia treated, dehydrated stepwise through alcohol, and cleared in xylene (148, 158). IHC detection of organisms proves infection, but negative does not rule out the disease.
Giemsa Staining
Smears or paraffin embedded slide sections stained withGiemsa reveals a fine nuclear detail of Toxoplasma organisms. It stains nuclei of the Toxoplasma organisms and permits differentiation among the cells (149).
Polymerase Chain Reaction (PCR)
Amplification of Toxoplasma DNA is preferred technique to diagnose congenital and disseminated toxoplasmosis in HIV/AIDS patients. PCR is applied to body fluids including blood, saliva, urine, CSF, bronchoalveolar lavage as well as brain tissue. Sensitivity may be variable, but specificity remains high. The real-time PCR assay has been shown to be the best-performing technique to detect DNA in amniotic fluid with 98% sensitivity and 100% specificity compared to other conventional PCR and nested PCR, or multiplex-nested-PCR techniques similarly utilizing B1 gene primers (198).
Pathology
The visualization of tachyzoites in infected tissue specimens with IHC stain or Geimsa stained smears of body fluids (amniotic fluid, CSF, bronchoalveolar fluid) along with infiltration of inflammatory cells can establish the diagnosis of acute infection.
Radiologic Examinations
CT and MRI scans showing calcifications in the brain or multiple lesions are major diagnostic modalities suggesting the presence of Toxoplasma encephalitis. The other major differential diagnostic possibility is CNS lymphoma. Brain biopsy may be indicated in a patient with sporadic lesion (31, 139).
Fetal Maternal Toxoplasmosis Diagnostics
Toxoplasma infection during pregnancy causes a serious health risk for the fetus. Thus, quick and accurate diagnostics are essential for possible preventions and available therapeutics. Fetal maternal toxoplasmosis is diagnosed using serological assays to detect types and levels of anti-Toxoplasma antibodies. Sera exam is necessary to be repeated during the gestation period and suggested at the birth. Therefore, pregnant women are routinely tested in Austria, France, Italy, Portugal, and Uruguay for antibodies detection. In addition, limited screening program are utilized in Belgium, Germany, and Switzerland. Fetal and neonatal screening for congenital toxoplasmosis are performed on more than two million pregnant women each year in Europe, and North and South America with estimated cost of more than 500 million dollars annually (150, 165). However, United States so far does not require routine screening of pregnant women for toxoplasmosis. Considering significant variation of the disease manifestation and cosmopolitan distribution of Toxoplasma infection, it is recommended that infants with unexplained severe systemic disease to be tested for congenital toxoplasmosis (9). Seronegative pregnant women signifying no previous infection are at risk for infection and recommended to be serologic tested monthly until birth.
Standard laboratory diagnosis of toxoplasmosis is based on the presence of IgM and IgG anti-Toxoplasma antibodies, and molecular technology is utilized as alternative tools to increase sensitivity (96). Typical infection is associated with high anti-Toxoplasma IgM antibody followed by a rise in IgG levels in 1 to 3 weeks after infection. However, atypical infection with IgG sero-conversion occurs in pregnancy without detectable IgM levels. These cases are very difficult to diagnose in congenital toxoplasmosis (72). The pregnant women with low or steady levels of IgG anti-Toxoplasma antibody have been previously exposed to the organism and fetus may remain safe. However, detection of IgM or elevation of IgG anti-Toxoplasma antibodies suggest a new acquired or reactivation of chronic/latent infection due to possible immunosuppression with a risk of infection for the fetus. Therefore, amniotic fluid exam can be performed to determine health status of the fetus and possible exposure to the infection. Toxoplasma-antibodies in infants during the first year of life present congenital toxoplasmosis.
In serious cases of neurological disorders including visual and auditory impairments, IgM and IgG antibodies combined with CSF polymerase chain reaction (PCR) is recommended to detect Toxoplasma DNA. Of 58 infants (0 to 180 days) born from infected moms, CSF PCR was positive in 46.5% and 100% negative in uninfected healthy infants. PCR was positive in 80% of those infants with hydrocephalus, 53% in cerebral calcifications and 60% of those with eye complications. Therefore, infants with neurological disorders, IgM and IgA antibodies combined with CSF PCR, are considered to yield a higher sensitivity for diagnosis of congenital toxoplasmosis when compared with each test alone (147).
Anti-Toxoplasma IgG and IgM with similar specificity and sensitivity are detected with the Vidas (bioMérieux), Architect (Abbott), and Liaison (DiaSorin) systems. The LIAISON system is a fully automated system based on chemiluminescence and antigen bound to magnetic microparticles. The Vidas system fits the needs of smaller laboratories with high specificity and a powerful avidity test in pregnant women and infants sera. A confirmatory serologic test is recommended to improve the rate of detection of congenital toxoplasmosis at 1 year of life (141, 164).
Additional serological tests to detect Toxoplasma specific IgG and IgM in blood samples are the indirect fluorescent antibody test (IFAT), microparticle enzyme immunoassay (MEIA) and enzyme-linked fluorescent assay (ELFA) used according the manufacturer’s instructions. The IgM sensitivity is reported 60% with specificity 100% except for IFAT with 92% and ELISA-IgA of 57% respectively (10). The molecular diagnostic include Western Blot analysis to detect anti-Toxoplasma IgM and IgA in infected infants (49). Cerebral toxoplasmosis is fatal if not treated and difficult to diagnose. Detection of anti-Toxoplasma IgM in CSF is reported as a reliable marker for cerebral Toxoplasmosis (96).
PATHOGENESIS
Following ingestion of the tissue cyst or oocyst form by humans, gastric digestive juices disrupt their outer cyst wall releasing infective forms, bradyzoites and sporozoites which rapidly invade intestinal enteroepithelial cells. They transform to active and replicate form of tachyzoites and infect adjacent cells to reach the lymphatic and blood stream. Organisms infect all nucleated cells and further invade brain, eye, heart and skeletal muscle and fat tissues as well as the placenta and fetal tissue to result congenital infection. Oral gavage of cysts containing bradyzoites resulted in inconsistent prototype for dissemination. Luciferase-derived in vivo signal imaging indicates significant numbers of tachyzoites are first observed in chest area and later signal spread to other organs and abdominal area. In contrast, mice infected via direct ingestion of infected organs, the signals from organisms are first detected replicating in the abdominal area (21).
Tachyzoites trigger a strong immune response responsible for the clinical symptoms of toxoplasmosis during the acute or reactivation of the latent infection. In immunocompetent individuals free tachyzoites provoke a strong immune system and are killed by serial activation of complement associated antibodies, reactive oxygen and nitrogen radicals, osmotic fluctuations, and intracellular acidification. Humoral immune responses, IgG, IgM, and IgA are responsible for lysing extracellular tachyzoites. However, some tachyzoites attach cells and invade intracellular milieu and parasitophorous vacuole to avoid the innate and adaptive immune mechanisms. These tachyzoites transform into bradyzoites with distinct structure and slower metabolism which replicate to form the resistant tissue cysts. Tissue cysts primarily form in muscles, heart, brain, and retina, are responsible for the chronic infection. In immunocompetent individuals the cell-mediated immunity response including macrophages, natural killer cells, CD4+ T cells (Th1) and CD8+ T cells are responsible to control the propagation of the organisms (2, 142). Cytokines such as TNFα, IFNγ, IL-1, IL-2, IL-4, IL-6, IL-7, IL-12, IL-15 mediate protective immunity. Co-stimulatory molecules such as CD28 and CD40 ligand are critical for the regulation of IL-12 and IFNγ production in response to the parasite. It is possible that different cytokines production initiates the extent of the infection in individuals. Hence, Toxoplasma may specifically impact levels of circulating cytokines, and results in differences in the clinical severity of toxoplasmosis (163). Tissue-specific immune responses, such as that in the central nervous system are being currently elucidated.
Immunosuppression caused by T-cell mediated immunity impairment as in administration of anti-CD52 monoclonal antibody, in AIDS, allogeneic hematopoietic bone marrow and transplantation may lead to the transformation of bradyzoites to the rapid dividing tachyzoites and reactivation and dissemination of the toxoplasmosis. Tachyzoites cause significant pathology in severely immunocompromised individuals with as high as 100% mortality rate if untreated. While in the presence of intact innate, humoral and cellular immune responses the acute infection eventually transition into the life-lasting chronic and asymptomatic phase waiting for an opportunity to become activated.
TOXOPLASMA GENOTYPES
The organism genetic analysis obtained from humans and animals mainly in North America and Europe has categorized Toxoplasma into three genetically distinct clonal lineages (types I, II, III) which may reveal important biological and clinical differences (105, 132, 184, 189). For instance, strains differ in a number of phenotypes, such as virulence, persistence, oral infectivity, and migration capacity, induction of cytokine expression, modulation of host gene expression and severity of disease. Therefore, the outcome of toxoplasmosis in patients becomes also variable (132). Type I strains demonstrate more acute and a greater ability to migrate and disseminate as well as more virulence than type II strains. Type I and II strains are significantly more prevalence and associated with disease in humans. Type III strains are more common in animals but less frequent observed in cases of human toxoplasmosis (92). In Europe, chronically infected pregnant women are predominantly infected with type II and in South America (Colombia) with types I and III (166). Recent genetic investigations in North America reveals most outbreaks result from contaminated water and vegetables with oocysts in cats’ feces and transmitted to humans and animals. The National collaborative Chicago-based congenital toxoplasmosis reported 78% of the mothers from epidemic area acquired primary infection from oocyst forms, while only 49% kept house cats (22, 150).
CURRENT AVAILABLE ANTI-TOXOPLASMA THERAPIES
Toxoplasma infection in healthy immunocompetent individuals is hardly detected and or treated. The related symptoms are usually resolved in a few weeks or months. However, treatments are required in unresolved (after 2 to 4 weeks) or severe cases as well as, in immunosuppressed and HIV/AID and congenital toxoplasmosis.
Pyrimethamine (Daraprim)
Antimicrobial pyrimethamine, a folic acid antagonist which inhibits the dihydrofolate reductase (DHFR), is a major enzyme in the purine pathway in organism (108). Pyrimethamine is effective against tachyzoites in acute toxoplasmosis but has no effect on cysts in chronic stage of disease. In addition it has cytotoxity effects as shown in vitro (43, 56) and in animal studies (168).
Pyrimethamine should be administered in combination with other effective drugs as the efficacy is very limited. The possibility of relapses following discontinuation of the drug in immunosuppressed patients warrants prolonged duration of treatment following the acute phase of the infection. Pyrimethamine is metabolized in liver and its pharmacokinetics is not altered by renal insufficiency. The lack of correlation between serum concentrations and varying pharmacokinetics between patients suggest genetic differences in metabolizing the drug and monitoring serum levels of pyrimethamine can be useful in patients.
Oral administration daily is necessary treatment in acute infection. Pyrimethamine in immunocompetent adults at a daily dose of 25 mg (0.3 mg/kg) leads to serum concentrations of 0.9 to 1.7 µg/ml. In HIV patients a dose of 50 to 75 mg/day leads to serum concentrations of 1 to 4.5 µg/ml with peak levels at 3.3 hours (105). A minimal serum concentration of 3.0 µg/ml may be required to treat Toxoplasmaencephalitis in HIV infected patients (212). Serum concentration 4 h after a daily dose of 1 mg/kg in infants was 1.3 µg/ml which decreased to 0.7 µg/ml if was given every other day (131) which remained fairly stable during treatment period. Concentrations of pyrimethamine achieved in infants' sera with standard dosages had effective inhibitory effect in vitro on most virulent strains including the RH strain (131). CSF concentrations of pyrimethamine range 10 to 25 % of the serum in patients with leukemia or AIDS patients with Toxoplasma encephalitis (212). Half-life of the drug was 40 hours in brain tissue and 28 hours in serum, which implies that a dose of 50-100 mg every other day is convenient for prophylactic therapy in AIDS. Pyrimethamine may interact with other drugs including phenobarbital affecting hepatic enzymes and the drug metabolism can be altered in liver disease patients.
Anti-Toxoplasma activity of sulfonamides is by inhibiting dihydrofolate-synthetase enzyme essential in the purine pathway in Toxoplasma, while have minor cytotoxicty and side effects (1,122). Sulfadiazine is commonly prescribed while other sulfonamides, sulfapyrazine, sulfamerazine and sulfadimidine are also effective and can be used as trisulfapyrimidine combination (sulfadimidine, sulfamerazine and sulfadiazine).
Spiramycin
A macrolide antibiotic, spiramycin is fairly safe and acts on Toxoplasma by inhibiting protein synthesis demonstrated in vitro and in murine model for toxoplasmosis. It is exclusively used to prevent maternal-fetal transmission of organisms. However spiramycin monotherapy is less effective than pyrimethamine or sulfadiazine. Spiramycin is primarily used in pregnant women to preventToxoplasma fetal maternal transmission in those with active or acquired Toxoplasma infection in early gestation period (≤18 weeks). It is mainly to prevent transmission of infection and not effective after transplacental transmission of organisms (76). Spiramycin is used broadly in Europe (76). Spiramycin has no teratogenic effect and is well-tolerated to be administered for several months.
A clinical trial in pregnant women treated with a daily dose of 3 g, the maternal serum, cord blood and placenta tissue average concentrations were 1.9 µg/ml, 0.8 µg/ml and 6.2 µg/g respectively (74). The fetal-maternal ratio in sera was about 0.5 by the weeks 21-24 of pregnancy and 0.7 at birth (64). Administration of Spiramycin to infected pregnant women is associated with a significant decrease (60%) of the risk for placental and fetal infection (43) and maternal-fetal transmission (47). Spiramycin is rapidly adsorbed by sera, with elevated levels to persist in tissues and placenta. It is ineffective against cerebral toxoplasmosis in AIDS patients (112). Spiramycin application is specifically to protect fetus against acquired toxoplasmosis during pregnancy in a dose of 3 g/day oral route in 2 or 3 divided doses [206].
Clindamycin
The effect of clindamycin on Toxoplasma has been established in murine models of acute and chronic toxoplasmosis (7). Clindamycin is absorbed following oral administration with peak of 4 and 8 µg/ml after ingestion of 300 and 600 mg tablets respectively (116) and a half-life of about 2.7 hours. It is excreted in urine and bile, and partially metabolized in active and inactive metabolites. Approximately 90 % of Clindamycin is bound to plasma proteins and dispersed in retina, iris and choroid and retina, but CSF and brain penetration is not proven. The efficacy of clindamycin plus pyrimethamine has been shown to be comparable to that of pyrimethamine plus sulfadiazine in AIDS patients with Toxoplasma encephalitis (39).
Other Macrolides
The inhibitory activity of roxithromycin, clarithromycin, and azithromycin has been observed in vitro in high concentrations but the mechanism of action is not well known (7, 8, 85). Roxithromycin is protective in murine with acute toxoplasmosis and high virulent strain of RH Toxoplasma but does not protect against cyst forming in the brain (8, 33).
Atovaquone
Atovaquone (566C80, hydroxy-1,4-naphthoquinone) is FDA approved in Adults toxoplasmosis (93). It is active in vitro and in vivo (179) and the most active compound against tachyzoites and cysts, yet not approved for fetal–maternal and children toxoplasmosis (35). Atovaquone is anti-fungal Pneumocystis pneumonia and anti-Babesia microti, causative of human blood-borne babesiosis endemic in New England and Northeastern of United States (94, 95, 159). Atovaquone acts by targeting mitochondrial respiration to block and collapse the membrane in the organisms (71, 193). Atovaquone has a half-life of 1.5–3 days and mainly binds to plasma proteins (99%) and is excreted into feces (94%) without being metabolized (177). Atovaquone has been shown to protect against maternal congenital toxoplasmosis and inflammatory complications in animal model (149, 158).
Atovaquone (administered orally as a suspension) combined with either pyrimethamine or sulfadiazine as treatment for acute disease for patients with Toxoplasma encephalitis has been shown to be effective, with 6-week response rates of 75% (21/28 patients) for atovaquone plus pyrimethamine and 82% (9/11) for atovaquone plus sulfadiazine. Thus, atovaquone plus pyrimethamine can be used as an alternative treatment for patients intolerant to sulfonamides, and atovaquone plus sulfadiazine for patients who are intolerant to pyrimethamine (34,107). In addition, combined atovaquone plus pyrimethamine or sulfadiazine are also effective in for Toxoplasma encephalitis in AIDS patients (34).
Trimethoprim, Trimetrexate, Piritrexime
Folate Inhibitors with mechanism of action similar to pyrimethamine to inhibit DHFR are reported to be effective in vitro and in animal models againstToxoplasma and other opportunistic disease Pneumocystis pneumonia (79, 156). Trimethoprim plus sulfamethoxazole combined regimen (trimethoprim 10 mg/kg/divided in two doses) has reported to have similar efficacy as pyrimethamine plus sulfadiazine in AIDS patients with Toxoplasma encephalitis (27, 108, 191, 201, 202).
Other Drugs
Combined dapsone (100 mg/kg/day) plus pyrimethamine (18.5 mg/kg/day) protected 100% of infected mice with no relapses after discontinuation of the treatment (42). Qinghaosu (artemisinin) extracted from a Chinese herb and its derivatives have in vitro activity against tachyzoites of Toxoplasma [8] as well as malarial organisms and herpes viruses.
Immunomodulators
The role of cellular immunity in the defense against Toxoplasma has been clearly established. Macrophages play an essential role in early immune response against infection. Recent findings indicate that IFNβ production by infected cells is associated with organisms’ death, independent of IFNγ activation. This action is dependent on endosomal Toll-like receptors in macrophages and the cytoplasmic receptor retinoic acid-inducible gene 1 (RIG-I) in fibroblasts (132). Additionally, IFNγ is effective in vitro and in vivo possibly by activating macrophages and stimulating natural killers and humoral response (128) and interfering in the metabolism of Toxoplasma. IFNγ was effective in mice with chronic Toxoplasma encephalitis (196) and protected against lethal infection (85). Combined IFNγ plus roxythromycin demonstrated synergistic effects in murine encephalitis (85). Recombinant interleukin 2 significantly decreased mortality in mice by reducing the number of cysts in brain [188]. IL-12 improved survival of T cell deficient mice during Toxoplasma infection by enhanced production of IFNγ from natural killer cells (75). The combined IL-12 plus clindamycin or atovaquone has been shown to be effective in murine toxoplasmosis well (5).
Combination Drugs
Pyrimethamine plus sulfonamides have synergistic effects to increase pyrimethamine activity 6-8 fold against tachyzoites (200) as demonstrated in animal models (168). In vitro studies reveal, pyrimethamine in 0.02 µg/ml and sulfadiazine 0.1 µg/ml to have synergism (43) versus inhibitory IC50 for sulfadiazine monotherapy to be 2.5 µg/ml. A minimal serum concentration of pyrimethamine 0.75 µg/ml in the presence of sulfonamides is needed to treat Toxoplasma encephalitis in HIV infected patients (212). In infants serum concentration at 4 h after a daily dose of 1 mg/kg was 1.3 µg/ml which decreased to 0.7 µg/ml when was given every other day (131).
Combined pyrimethamine plus sulfadiazine with proven synergistic effect remains as basis treatment for human toxoplasmosis, but possible side effects including rash creates major obstacle for application. Pyrimethamine plus sulfadoxine can be used for prolong treatment and available in oral or parenteral route. In case of rash desensitization program is practiced (172). Combined pyrimethamine plus sulfadoxine has been shown with synergistic effect in murine toxoplasmosis (122), while 100% of organisms were eradicated when treated immediately and only 32% when delayed 72 hours after inoculation of organisms. Pyrimethamine plus sulfadoxine has severe side effects.
Other Combinations
Combined pyrimethamine plus clindamycin has been shown to be effective in AIDS patients with Toxoplasma encephalitis (39). Similarly combination of trimethoprim plus sulfamethoxazole (201, 202), pyrimethamine plus atovaquone or atovaquone plus sulfadiazine have been demonstrated to be effective as well in the AIDS population (34, 107). Oral administration of trimethoprim plus sulfamethoxazole and intravenous clindamycin combined with short-term dexamethasone for the cerebral edema, have been reported effective against cerebral toxoplasmosis in patient with improved symptoms and reduced antibodies (96). Combined clarithromycin plus sulfadiazine is reported with high synergistic effect (6).
THERAPEUTIC INDICATIONS
Acute Toxoplasmosis in Immunocompetent Patient
Immunocompetent patients with severe symptomatic disease including, lymphadenopathy, myositis, myocarditis, hepatitis, or ocular involvement require treatments.
Acute Infection During Pregnancy
Spiramycin generally is recommended to manage fetal maternal infection for the first and early second trimesters. Spiramycin can reduce the risk of maternal-fetal transmission when treatment started before infection has reached the embryo. Spiramycin in U.S.A is available through the Investigational New Drug (IND) process at the Food and Drug Administration [FDA, Administration (301/796-1600). Spiramycin is not recommended when fetus is suspected to be infected (see below recommendations for pyrimethamine plus sulfadiazine and folinic acid).
Application: Spiramycin is recommended for pregnant women suspected acquired the infection < 18 weeks of gestation and administered until delivery.
Spiramycin (PO) dose: 1g (3 million units)/8 hr or total: 3g (9 million units)/day
Alternatively pyrimethamine plus sulfadiazine and leucovorin is given to pregnant women in the late second and third trimesters to treat positive infected fetus by means of PCR for amniotic fluid or suspected with abnormal ultrasound after the 18 week of pregnancy following written consent and under close supervision (138, 139). Pyrimethamine plus sulfadiazine passes placental barrier and is recommended for women with acquired acute infection after 18 weeks of gestation. Due to the increased risk of maternal-fetal transmission of the organisms in late pregnancy indicated (180). Pyrimethamine 50 mg/day and sulfadiazine 3g/day is administered until delivery with hematological monitoring. Pyrimethamine plus sulfadiazine significantly reduce the rate of Toxoplasma isolation from the placenta and antibody synthesis of specific IgM and IgG titers in the neonate. Side effects include fetal injury. Trimethoprim plus sulfamethoxazole has been recommended as an alternative to treat congenital toxoplasmosis during pregnancy (45).
Recommendation: Pyrimethamine is teratogenic and should not be used in pregnancy before week 18.
Recommended for women suspected of having acquired the infection at ≥ 18 weeks of gestation; those with positive amniotic fluid PCR test or abnormal ultrasound suggestive of congenital toxoplasmosis, and who is over 18 weeks of gestation.
Application: Pyrimethamine: 50 mg/12 hr for 2 days followed by 50 mg daily (PO)
Sulfadiazine: 75 mg/kg (first dose) followed by 50 mg/kg every 12 hr (maximum 4 g/day) (PO)
Leucovorin (Folinic acid): 10-20 mg daily (during and for 1 wk after pyrimethamine therapy) (PO)
Congenital Toxoplasmosis
Infants with congenital toxoplasmosis require therapy during the first year of life in order to treat acute disease or to prevent secondary complications (172). About 75% of infants which were exposed to infection during gestation period from moms treated with spiramycin develop latent disease. Postnatal treatments are necessary when fetus diagnosed positive by means of amniotic fluid, specific and IgM or IgA seropositive for active infection after birth or Toxoplasma isolated from placenta. In contrast only 10% of infants may have negative isolation when their infected mother was not treated, 25% from mother treated with spiramycin and 50% from mothers received spiramycin and pyrimethamine plus sulfadiazine combined with leucovorin as typical drugs in alternation therapy for 1 year (36, 139). Therefore, infants with negative PCR in the amniotic fluid and negative results for organisms in placenta still require treatment to prevent congenital toxoplasmosis.
Pyrimethamine plus sulfadiazine is the standard treatment to prevent ocular symptoms. The Chicago Collaborative Treatment Trial recommendation requires continuous therapy for the first year of life in those children with possible infection (127). Acute retinochoroiditis has been shown to become subclinical within two weeks or less following the application of therapy and without visual loss in most treated patients (133). Most infants without hydrocephaly have normal IQ ranges versus half of those with hydrocephaly (127, 176, 197). About 75% of infants treated early after birth for toxoplasmosis, intracranial calcifications diminished or resolved by age 1 (160). While serologic titers appeared in 97.7% of the cases following discontinuation of the treatment, but this did not lead to an increased risk of ocular relapse (50). A clinical trial in 257 newborns from infected moms treated with spiramycin and pyrimethamine plus sulfadoxine in France, 24% were diagnosed with congenital infection. From these, 7% were predicted to be infected in the first, 24% in second, and 59% in third gestation period, respectively (17). Infected infants are required to be treated for 1 year.
Application: Pyrimethamine: 1 mg/kg /12 hr for 2 days, followed by 1 mg/kg/day for 2- 6 months, followed by 1 mg/kg three doses/week
Sulfadiazine: 50 mg/kg/12 hr
Leucovorin (Folinic acid): 10-20 mg three times/ week
Prednisone (CSF protein ≥ 1 g/dL for retinochoroiditis): 0.5 mg/kg/12 hr until CFS protein < 1 g/dL or symptoms resolve.
Treatment of older children with active disease continues 1-2 weeks after resolution of symptoms.
Pyrimethamine: 1-25 mg/kg every 12 hr for 2 days followed by 1-25 mg/kg per day
Sulfadiazine: 75 mg/kg (first dose) followed by 50 mg/kg every 12 hr
Leucovorin (Folinic acid): 10-20 mg three times weekly
Prednisone for retinochoroiditis: 0.5-20 mg/kg/12hr, rapid taper
Ocular Toxoplasmosis
Acute ocular toxoplasmosis requires urgent therapy for 6 to 8 weeks and a minimum of 2 weeks after inflammation is resolved in ophthalmic and angiographic examination. Commonly, in 2 weeks of treatment ocular lesions are resolved and further visual loss seized (125, 133). Pyrimethamine (100 mg for 1 day followed by 25 to 50 mg/day) is used plus sulfadiazine (1g four times/day), plus leucovorin(folinic acid, 5-25 mg). Pediatric: pyrimethamine (2 mg/kg first day then 1 mg/kg/day) plus sulfadiazine (50 mg/kg twice/day), plus leucovorin (7.5 mg/day) for 4 to 6 weeks followed by reevaluation of the patient. Leucovorin is used to protect the bone marrow from toxic effects of pyrimethamine. In case of hypersensitivity reaction to sulfa drugs, pyrimethamine plus clindamycin is recommended. In contrasttrimethoprim plus sulfamethoxazole combination has been used in case of pyrimethamine intolerance. Other alternative drugs are atovaquone plus pyrimethamine and azithromycin (31, 125, 161). Steroids (corticosteroid) are recommended as adjuvant for patients with severe inflammatory responses, retinochoroiditis and macula involvement or CSF protein ≥ 1 g/100ml (73).
Infection in Immunocompromised Patient
In immunosuppressed patients, treatment of acute toxoplasmosis is required to protect against 100% mortality rate. Therapies and duration depend upon clinical manifestation, oral administration, severity and duration of the immunosuppression (190). Maintenance therapy is modified according to the duration and the intensity of the immunosuppression. Similar criteria are used as in HIV and AIDS patients. Usually it is discontinued in transplant recipients after the immunosuppressive agent is tapered.
Infection in AIDS Patients
The number of cases of toxoplasmosis in HIV infected patients has remarkably decreased primarily due to the extensive use of trimethoprim plus sulfamethoxazole as primary prophylactic regimen for bothPneumocystis pneumonia (156) and toxoplasmosis, as the use of highly active antiretroviral therapy (HAART) lead to immune restoration. The recommendations are based on data from trials conducted before 1996 (204). Persons with AIDS who develop active toxoplasmosis including Toxoplasma encephalitis should be treated until significant immunologic improvement is achieved (73).
STANDARD OF CARE THERAPY
Acute Therapy
The therapy of choice for Toxoplasma encephalitis consists of the combined pyrimethamine plus sulfadiazine and leucovorin (folinic acid) (69,144). Pyrimethamine penetrates to bypass brain blood barrier efficiently even in the absence of inflammation. Leucovorin reduces the possible hematologic toxicities associated with pyrimethamine therapy (69). The dose can be increased up to 50 mg/day in case of cytopenia. The duration of acute stage therapy is 3-6 weeks. Only if a complete clinical and radiological response is obtained therapy can be concluded (111, 210). A complete or partial clinical response to the combined therapy is achieved in 70% to 75% cases of Toxoplasma encephalitis in HIV-infected patients with probability of survival after an acute episode to be 90% to 95% (39). The combined therapy has similar rate for other sites of infection such as the eyes. A disseminated infection followed by the septic shock leads to a poor prognosis (118).
Trimethoprim plus sulfamethoxazole was reported in a small (77 patients) randomized trial to be effective and better tolerated than pyrimethamine plus sulfadiazine (201). No parenteral formulation of pyrimethamine exists for the patients who cannot take oral tablets. However, sulfamethoxazole is available in parenteral form. ICU patients and those with severe Toxoplasma encephalitis can receive parenteral trimethoprim plus sulfamethoxazole or oral pyrimethamine plus parenteral clindamycin.
Treatment is usually initiated upon assumption of Toxoplasma encephalitis, based on clinical symptoms of fever, neurological manifestations, seizers and the presence of brain lesions detected by CT scan or MRI. The diagnosis of Toxoplasma encephalitis is less likely in Toxoplasma seronegative patients (e.g. HIV+) with single lesion detected by MRI and/or patients who are on trimethoprim plus sulfamethoxazole prophylactic for Pneumocystis pneumonia (31, 120, 156).
Application: Pyrimethamine 50 mg/12 hr (2 days) followed by 25-50 mg daily
Leucovorin (Folinic acid) 10-50 mg/day (PO) continued 1 week after Pyrimethamine.
Sulfadiazine (PO) 75 mg/kg (first dose) followed by 50 mg/kg/12hr
Trimethoprim plus Sulfamethoxide 5-10 mg/kg/12 hr (PO or IV)
Maintenance Therapy
To prevent relapse maintenance therapy has been well established practice in AIDS patients (80). The relapse rate is reported approximately 30% despite zidovudine therapy (40). Maintenance therapy is based on the combined therapies used at doses reduced by half. The combined therapy with pyrimethamine plus Sulfadiazine has been shown to be more effective than the pyrimethamine plus clindamycin alternative combined therapy to prevent relapse. European study revealed relapse rate to be 6% patient with sulfadiazine versus 23% patient with clindamycin (104). Maintenance therapy can be discontinued in patients with CD4 counts above 200 cells/µl, and undetectable HIV viral load for 3 to 6 months.
Alternative Therapeutic Regimens
The alternative therapy for patients, who do not tolerate or fail to respond, is combined pyrimethamine plus clindamycin and leucovorin (173). About 70% - 86% of the patients had a favorable response and improved by 1-6 week of therapy. The adverse effects included non-Toxoplasma related neurological symptoms due to the pyrimethamine plus clindamycin combined therapy in 30-50% of patients, which required discontinuation in 20% cases. The adverse effects of clindamycin are primarily gastrointestinal due to microbiota alteration with a risk of Clostridium difficile colitis. The combined therapy can be used in half reduced dose for the maintenance therapy to prevent side effects but can also lower efficacy compared the conventional therapeutic doses (104).
Combined therapies with trimethoprim plus Sulfamethoxazole, Pyrimethamine plus Atovaquone or Ataovaquone plus Sulfadizine are also effective in AIDS patients with Toxoplasma encephalitis.
Combined Pyrimethamine plus Macrolides
Due to the high rate of adverse reaction and intolerance to above combinations, specifically in AIDS patients other combinations of pyrimethamine plus clarithromcyin (2g/day) or azithromycin (500 mg/day) might be effective (60). The adverse effects requiring discontinuation in 20% of patients were increased in serum transaminase levels and hearing loss in two studies. After successful use of combination therapy during the acute or primary stage, one half of doses of same agents are recommended for maintenance or secondary prophylaxis.
Application: Pyrimethamine 50 mg/12 hr (2 days) followed by 25-50 mg daily
Leucovorin (Folinic acid) 10-50 mg/day (PO) plus pyrimethamine and continued 1 week after
Sulfadiazine 75 mg/kg (first dose) then 50 mg/kg/12 hr (PO)
1000 (<60 kg) to 1500 mg (>60 kg) every 6 hr
Or Clindamycin 300-1200 mg/6 hr (PO or IV)
Atovaquone 1500 mg/12 hr (PO)
Trimethoprim plus Sulfamethoxide 5-10 mg/kg/12 hr (PO or IV)
Pyrimethamine/folinic acid
Plus Clarithromycin 500 mg/12 hr (PO)
Or Dapsone 100 mg/d (PO)
Or Azithromycin 900-1200 mg/day(PO)
Adjuvant Therapy
Congenital Toxoplasmosis
Fetal abortion is considered if fetal infection is documented with evidence of fetal abnormalities on ultrasound examination (16). In infants with latent infection treated with the combined pyrimethamine plus sulfonamides and prednisone (1mg/kg/day) oral route is administered in two divided doses, against inflammatory process, high cerebrospinal protein content, and severe uveitis. The dose is tapered when the disease progress subsided.
Ocular Toxoplasmosis
Corticosteroid is administered as adjuvant therapy for inflammatory condition. Prednisone or methylprednisone (80 mg/day) is given in adults and children (1.5 mg/kg/day) and dosage tapered. The primary anti-Toxoplasma agent will continue minimum 2 weeks after steroids is discontinued (111, 119).
AIDS
Adjuvant therapy with corticosteroids in Toxoplasma encephalitis is routinely administered in AIDS patients with clinical or radiological signs of brain edema. Solumedrol (240 mg/day) is prescribed for 3 days, followed by half dose (120 mg/day) for 3 additional days, and then tapered by half (60 mg/day) until significant clinical improvement (10 to 20 days). Other regimens such as glycerol or mannitol have been used in intracranial hypertension to reduce the symptoms (111, 121).
ENDPOINTS FOR MONITORING THERAPY
Monitoring of Adverse Events
Pyrimethamine administered in high doses may cause seizures. Children (25kg) receiving 2 mg/kg pyrimethamine or patients with serum levels of 5 µg/ ml and above are at risk (131) and require blood transfusion for detoxification. Pyrimethamine has antifolinic activity results in bone marrow depression, neutropenia, thrombocytopenia, and megaloblastic anemia. The side effects are prevented by oral administration of leucovorin (calcium folinic acid) 5 to 10 mg three times/week for infants and 10 up to 50 mg/day in adults and HIV infected patients. In fact, Toxoplasma cannot consume folates, folic and folinic acid, and requires to metabolize precursor paraminobenzoic acid. HIV infected patients required to be evaluated for neutrophil counts two times/week and platelets and hematocrit once/month (127). Neutrophil count below 1000/µl or platelets 90,000/µl necessitate to escalate folinic acid dosage or withdraw when neutrophils decrease 500/µl to reverse cytopenia. Leucovorin must be administered one week after pyrimethamine discontinued (69, 205).
Sulfonamides
Treatment with sulfonamides such as sulfadiazine requires monitoring the hematological cell count and blood creatinin, amount of urine output, urea crystal formation, and water consumption (37). Adequate hydration and alkalizing the urine can be utilized to prevent the side effects. In case of nephrolithiasis sulfonamides need to be discontinued. Trisulfamides can be administered as alternative with fewer side effects.
Pyrimethamine plus Sulfonamides Combination therapy
The rate of side effects due to the combined pyrimethamine plus sulfadiazine therapy is approximately 50%, and discontinuation of one or both drugs is required in 20% to 25% cases (39,104). The most frequent adverse effects include cytopenia, rash and fever. Alternative use of each drug will detect the drug responsible to be discontinued.
Clindamycin
The most frequent adverse effects are gastrointestinal manifestation, possibly due to alteration of microbiota and pseudomembranous colitis induced by Clostridium difficile. Other adverse effects seen in AIDS patients treated with combined clindamycin plus pyrimethamine, are rash, hepatic function anomalies, neutropenia and thrombocytopenia (39, 194).
EXPERIMENTAL THERAPIES
Promising Experimental Compounds
Other compounds, combined rifabutine plus atovaquone for 30 days or rifabutine plus clindamycin for 15 days showed synergism against Toxoplasma encephalitis in mice, significantly improved brain inflammation and prolonged survival but did not protect relapses (178). However, combined rifabutine plus pyrimethamine and rifabutine plus sulfadiazine had not significant effect on brain inflammation compared with each drug alone (8). Combined trimetrexate (37 mg/kg/day) plus sulfadiazine (375 mg/kg/day) prolonged survival (93%) against acute toxoplasmosis in mice. Dapsone plus sulfamethazine (100 mg/kg/day) with additive effects was active in vivo against organism.
Experimental Diclazuil, Atovaquone Monotherapy and Combination Therapy
Diclazuril [4-chlorophenyl [2,6-dichloro-4-(4,5-dihydro-3H-3,5-dioxo-1,2,4-triazin-2-yl)pheny l acetonitrile] is commonly used in poultry and livestock against coccidiosis. Diclazuril specifically affects apicoplast, a rudimentary organelle from Toxoplasma, and not present in humans and animals. Diclazuril is a nontoxic agent with rapid absorption following oral administration to reach a constant level in plasma and cerebrospinal fluid. Recent studies have shown diclazuril to be well tolerated and effective in murine model for maternal and congenital toxoplasmosis (148, 149, 157). Diclazuril has no teratogenic effect at the administered doses.
While atovaquone protects against some aspects of gastrointestinal complications in experimental congenital toxoplasmosis in murine (158), diclazuril was superior to atovaquone in improving anemia, colonic length and hepatic complications against maternal toxoplasmosis (157). In addition, diclazuril plus atovaquone combination therapy exerted a unique synergistic effect against more severe fetal maternal toxoplasmosis and superior action to diclazuril or atovaquone monotherapy (148, 149). Diclazuril and diclazuril plus atovaquone warrant clinical trials in maternal congenital as well as in ocular and chronic toxoplasmosis. Finally, Diclazuril is anticipated to be used as a novel protective and preventive measure to eliminate the cycle of Toxoplasma infection in the definitive host, feline.
VACCINES
An effective vaccine to protect cats against sexual stage and oocysts production and asexual stages and cysts production in animals and humans is desirable to eradicate the disease. Thus far, there is no safe and effective vaccine available against Toxoplasma infection. The only commercialized vaccine is an attenuated-live Toxoplasma (Toxovax®)for veterinary use with limited success (25). This live vaccine protects sheep from abortion due to infection but is not always effective. The other products have been candidate for vaccines include, rhoptry proteins, the major virulence factors and various DNA formToxoplasma yet to provide a limited or no protection. Irradiated tachyzoites given orally to 1 week pregnant rats did not provide protection in pups and dams against challenge inoculation of the organisms analyzed by quantitative PCR. However, immunization protected the birth rate and litter size (26).
PREVENTIVE MEASURES
Common sense and basic hygienic measurements and hands wash are required as the first step to prevent transmission of the Toxoplasma infection. Fruits and vegetables should be thoroughly cleaned or peeled prior consumption. Meat products and sea food need to be cooked appropriately or kept frozen at sub 0o degree temperature. Milk products need to be pasteurized and sanitary or treated water used for drinking. Vulnerable individual, mainly pregnant women, children and immunocompromised subjects should avoid direct contact with cat feces. Cats need to be kept indoors and fed cooked or commercial food. Protective gloves should be worn before handling contaminated material including sandboxes, garden soil, or raw meat and hands washed after handling and prior consumption.
PROPHYLACTIC THERAPEUTIC AGENTS
Congenital Toxoplasmosis
Management of seroconverters is determined by the risk of maternal-fetal transplacental transmission of Toxoplasma. The average risk of 40% is reduced by about one-half after spiramycin treatment (139). Spiramycin has been used in fetal-maternal toxoplasmosis treatment and prevention in Canada, Latin America and Europe for decades while still is considered as an “experimental therapy” in United States. Spiramycin monotherapy is effective in early pregnancy as a preventive measure but not after fetal exposure to the infection. In a prospective cohort clinical trial in Brazil 58% of neonates from mothers treated with spiramycin and over 73% of those from untreated had congenital infection (10). Over 50 percent of patients which were treated with spiramycin retained Toxoplasma DNA in peripheral blood and remained infected (83).
The date of maternal infection is of important relevance to transmission. Immunocompetent moms normally have no risk if had exposed before conception. However, infection during pregnancy period has a high risk of transmission and congenital toxoplasmosis as the later the infection date during pregnancy, the higher the risk of transmission (47). Among women with seroconversion, the incidence of maternal-fetal transmission increases as the pregnancy progresses. Spiramycin is most effective if given as prophylactic and before infection has reached the embryo. In pregnant women treated with spiramycin, the incidence of fetal transmission increases from 2%, if maternal infection happen during 3 to 10 weeks of gestation, to over 30% when after 31 weeks (87). The incidence reaches 90% in infections during the last two weeks of pregnancy. In addition, the earlier the maternal infection occurs during pregnancy, the higher the risk of brain injury in the fetus. It is indicated that maternal infection after week 26 may become more subclinical.
Prophylactic spiramycin is administered as 3g per day, oral route, and should be given immediately after detection of infection during pregnancy to reduce the possible transmission to the embryo (47, 65,66, 208) or to reduce the severity of the disease in possibly infected infant (67). A negative PCR in amniotic fluid requires spiramycin administration to prevent or delay maternal fetal transmission. Administration of azithromycin in 500 mg/day for 3 times/ week for 1 to 4 weeks showed Concentrations in placental tissue were 10 fold higher in placenta than in amniotic fluid or maternal or cord blood (194).
Ocular Toxoplasmosis
Repeated exacerbations of ocular toxoplasmosis raise the requirement for a prolonged prophylactic. The pyrimethamine plus sulfadoxine combination, 1 tablet for 20 kg every 10 days for 6-12 months or longer as needed can be prescribed and the clinical and hematological changes closely monitored (167).
AIDS
The prevalence of encephalitic toxoplasmosis in HIV patients is reported 30-40% in Europe, with mortality rate of 20% which has promoted specific preventive measures to control reactivation ofToxoplasma in seropositive patients (97, 204). In contrast the prevalence of primary infection in seronegative patients is as low as 2%. However, these patients should be monitored regularly forToxoplasma titers (1-2/year) [15]. Toxoplasma seropositive patients ought to be assessed for possible risk of Toxoplasma encephalitis, and prophylactic therapies (14, 73, 78, 110).
Toxoplasma Encephalitis in Seropositive Patients
Toxoplasma encephalitis occurs almost exclusively due to reactivation of latent tissue cysts (31). Primary infection less often is associated with acute cerebral or disseminated disease. The severity of the immune defect is a major factor. According CDC the stage B or C, clinical manifestations, and CD4+ cell count less than 100/µl are independent factors associated with an increased risk of Toxoplasmaencephalitis (31). The probability to develop Toxoplasma encephalitis in one year rises 24% in patients with CDC stage C manifestations and 20% in patients with CD4+ below 50/µl (46). Furthermore, the IgG anti Toxoplasma antibody titer determined by ELISA at baseline is a preferred prognostic marker for risk of Toxoplasma encephalitis (46). The risk increases by 3 fold in patients with titers >150 IU/ml (36, 101) for IgG by Western blot and is associated with an increased risk of Toxoplasma encephalitis (110).
Primary Prophylactic Therapies
Patients who are given trimethoprim plus sulfamethoxazole for Pneumocystis prophylactic have a very low incidence of Toxoplasma encephalitis (28, 84, 124, 169, 185), as trimethoprim plus sulfametoxazole is known prophylactic drug of choice (204) A prospective randomized trial in France to determine the most convenient dose of trimethoprim plus sulfametoxazole was single strength dose per day compared to double strength tablet per day. Some experts suggest an every-other day administration (175). The pyrimethamine plus dapsone combination is effective in preventing Toxoplasmaencephalitis (77). However the rate of cross-intolerance between dapsone and trimethoprim plus sulfamethoxazole is approximately 40% (102). In an international double blind placebo controlled study no significant difference was detect between pyrimethamine, 50 mg thrice weekly or placebo for primary prophylaxis of Toxoplasma encephalitis (109). Rash was the major adverse effect in pyrimethamine arm. In another trial increased mortality was reported in the arm with pyrimethamine compared to payrimethamine plus folinic acid (98). HIV positive patients who are receiving prophylactic trimethoprim plus sulfametoxazole for Pneumocystis have minimum risk of Toxoplasma encephalitis. The patients may not tolerate well trimethoprim plus sulfamethoxazole. Overall individuals should be assessed for clinical stage of HIV infection, the CD4 cell count, and the Toxoplasma antibody titers.
Toxoplasma Prophylaxis and Highly Active Antiretroviral Therapy
The world widespread administration of highly active antiretroviral therapy (HAART) since 1996 has significantly improved the risk of opportunistics including toxoplasmosis in HIV positive patients. HAART therapy significantly elevated CD4+ cell counts followed by restoration of the immune system and has reduced the rate of toxoplasmosis to about 5%. Thus, USPHS consensus panels recommend that primary prophylaxis to be discontinued for toxoplasmosis in patients with viral control and CD4+ greater that 200/µl for over 6 months. The possibility for discontinuing secondary prophylaxis has also been suggested (81, 204).
Prophylaxis for Other Immunosuppressed Hosts
Organ transplants recipients are in a great risk of toxoplasmosis (88, 121, 192, 215). Immunosuppressants especially corticosteroids significantly increase the risk of toxoplasmosis. Cyclosporine is reported to have some antiparasitic activity in vitro and animal experiments and recommended as preferred treatment in patients at risk for toxoplasmosis (129), others report fatal toxoplasmosis in conjunction with cyclosporine therapy (13). Mycophenolate mofetil (cellcept) commonly used in kidney transplant has strong therapeutic anti-fungal (Pneumocystis) and antimicrobial effect (155), but little information available regarding the anti-Toxoplasma efficacy of mycophenolate.
Those organ recipients, who are seronegative for Toxoplasma and transplanted organ from seropositive for Toxoplasma donor, are recommended to receive pyrimethamine 50 mg/day to markedly reduce the risk of toxoplasmosis (88, 121, 215). Trimethoprim plus sulfamethoxazole is a possible alternative which has not been extensively assessed. In bone-marrow recipients, the risk of toxoplasmosis is the highest in seropositive patients who receive bone-marrow from a donor who is seronegative for Toxoplasma (44, 63, 126, 146). The pyrimethamine plus sulfadoxine or trimethoprim plus sulfamethoxazole combinations have been recommended for children. In these transplant patients, the duration of chemoprophylaxis depends upon the duration of the immunosuppressive therapy. Limited information is available for other causes of immunosuppression (19). It has been suggested that patients with immune cell defects or those on long term corticosteroid therapy, should be considered for prophylaxis of toxoplasmosis when the CD4 cell count reaches less than 100/µl. Pyrimethamine or trimethoprim plus sulfamethoxazole should be recommended for the duration of the immune defect persists.
Additional Information
Consultation assistance is available for the diagnosis and management of patients suspected with toxoplasmosis:
Chicago, U.S.A National Collaborative Treatment Trial Study (NCCTS), telephone number (773) 834-4152.
The Toxoplasma Serology Laboratory (Dr Jack Remington), the Palo Alto Medical Foundation, Palo Alto, CA. Website: http://www.pamf.org/serology/; Phone number: 650 853 4828]
Centers for Disease Control and Prevention, Atlanta, GA, 1-800-CDC-INFO, weekday 8am-8pm EST. cdc.gov/parasite/toxoplasmosis.
Spiramycin (Rhone-Poulenc Pharmaceuticals) is not FDA approved, and available via individual investigator IND for treatment of pregnant women with toxoplasmosis. Information on the Spiramycin program is available by the Division of Special Pathogens and Immunologic Drug products at FDA at (301) 827-2335.
FDA Adverse Events Report
The Safe Medical Devices Act of 1990 (SMDA) requires hospitals and other user facilities to report deaths and serious illnesses and injuries associated with the use of medical devices.
Erroneous result or any adverse event from an anti-Toxoplasma commercial test kit resulting in death or serious illness must be reported.
Voluntary report is encouraged directly to MedWatch, the FDA's voluntary reporting program. Telephone at (800) FDA-1088, by FAX at (800) FDA-0178,
Mail to: MedWatch, Food and Drug Administration, Rockville, MD 20857
REFERENCES
1. Allegra CJ, Boarman D, Kovacs JA, Morrison P, Beaver J, Chabner BA, Masur H. Interaction of sulfonamide and sulfone compounds with Toxoplasma gondii dihydropropteroate synthetase. J Clin Invest 1990;85:371-379. [PubMed]
2. Amodio G1, Mugione A, Sanchez AM, Viganò P, Candiani M, Somigliana E, Roncarolo MG, Panina-Bordignon P, Gregori S. HLA-G expressing DC-10 and CD4(+) T cells accumulate in human decidua during pregnancy. Hum Immunol. 2013;74:406-11. [PubMed]
3. Andiappan H, Nissapatorn V, Sawangjaroen N, Chemoh W, Lau YL, Kumar T, Onichandran S, Suwanrath C, Chandeying V. Toxoplasma infection in pregnant women:a current status in Songklanagarind hospital, Southern Thailand. Parasit.Vectors2014;7:239. [PubMed]
4. Arantes TP, Lopes WD, Ferreira RM, Pieroni JS, Pinto VM, Sakamoto CA, Costa AJ. Toxoplasma gondii: Evidence for the transmission by semen in dogs. Exp.Parasitol. 2009;123:190-4. [PubMed]
5. Araujo FG, Hunter CA, Remington JS. Treatment with Interleukin 12 in Combination with Atovaquone or Clindamycin Significantly Increases Survival of Mice with Acute Toxoplasmosis. Antimicrob Agents Chemo 1997;41:188-190. [PubMed]
6. Araujo FG, Prokocimer P, Lin T, Remington JS. Activity of clarithromycin alone or in combination with other drugs for treatment of murine toxoplasmosis. Antimicrob Agents Chemother 1992;36: 2454-2457. [PubMed]
7. Araujo FG, Remington JS. Effect of clindamycin on acute and chronic toxoplasmosis in mice. Antimicrob Agents Chemother 1974;6:647-51. [PubMed]
8. Araujo FG, Suzuki Y, Remington JS. Use of rifabutin in combination with atovaquone, clindamycin, pyrimethamine, or sulfadiazine for treatment of toxoplasmic encephalitis in mice. Eur J Clin Microbiol Infect Dis 1996(b);15:394-397. [PubMed]
9. Armstrong L, Isaacs D, Evans N. Severe neonatal toxoplasmosis after third trimester maternal infection. Pediatr Infect. Dis. J.2004;23:968-9. [PubMed]
10. Avelino MM, Amaral WN, Rodrigues IM, Rassi AR, Gomes MB, Costa TL, Castro AM. Congenital toxoplasmosis and prenatal care state programs. BMC Infect. Dis. 2014;14:33. [PubMed]
11. Bachmann S, Schröder J, Bottmer C, Torrey EF, Yolken RH. Psychopathology in first-episode schizophrenia and antibodies to Toxoplasmagondii. Psychopathology 2005;38:87-90. [PubMed]
12. Bahia-Oliveira LM, Jones JL, Azevedo-Silva J, Alves CC, Orefice F and Addiss DG. Highly endemic, waterborne toxoplasmosis in north Rio de Janeiro state. Brazil. Emerg Infect Dis 2003;9:55-62. [PubMed]
13. Barrs VR, Martin P, Beatty JA. Antemortem diagnosis and treatment of toxoplasmosis in two cats on cyclosporintherapy. Aust Vet J. 2006;84(1-2):30-5. [PubMed]
14. Beaman MH, Luft BJ, Remington JS. Prophylaxis for toxoplasmosis in AIDS. Ann Intern Med 1992;117:163-164. [PubMed]
15. Belanger F, Derouin F, Grangeot-Keros L, Meyer L and the HEMOCO-SEROCO study group. Incidence and risks factors of toxoplasmosis in a cohort of human immunodeficiency virus-infected patients: 1988-1995. Clin Infect Dis 1999;28:575-581. [PubMed]
16. Berrebi A, Kobuch WE, Bessieres MH, Bloom MC, Rolland M, Sarramon MF, Roques C, Fournie A. Termination of pregnancy for maternal toxoplasmosis. Lancet 1994;344:36-39. [PubMed]
17. Bessières, MH, Berrebi A, Cassaing S, Fillaux J ,Cambus, JP, Berry A, Assouline C, Ayoubi JM, Magnaval JF. Diagnosis of congenital toxoplasmosis:prenatal and neonatal evaluation of methods used in Toulouse University Hospital and incidence of congenital toxoplasmosis. Mem. Inst. Oswaldo Cruz2009;104:389–392. [PubMed]
18. Bosh-Driessen EH, Rothova A. Recurrent disease in potentially acquired toxoplasmosis. Am J Ophtalmol 1999;128:421-425. [PubMed]
19. Bouree P, Romand S, Majoube A. Toxoplasmose et transplantation hepatique : surveillance de 34 patients par deux techniques serologiques. Bull Soc Fr Parasitol. 1995;13:155-160.
20. Bowie WR, King AS, Werker DH, et al. Outbreak of toxoplasmosis associated with municipal drinking water. Lancet 1997;350:173-177. [PubMed]
21. Boyle JP, Saeij JP, Boothroyd JC. Toxoplasma gondii: inconsistent dissemination patterns following oral infection in mice. Exp Parasitol. 2007;116:302-5. [PubMed]
22. Boyer K, Hill D, Mui E, Wroblewski K, Karrison T, Dubey JP, Sautter M, Noble AG, Withers S, Swisher C, Heydemann P, Hosten T, Babiarz J, Lee D, Meier P, McLeod R; Toxoplasmosis Study Group. Toxoplasmosis study group. Unrecognized ingestion of Toxoplasma gondii oocysts leads to congenital toxoplasmosis and causes epidemics in North America. Clin Infect Dis. 2011;53:1081–1089. [PubMed]
23. Brandtzaeg P. Inflammatory bowel disease: Clinics and pathology. Do inflammatory bowel disease and periodontal disease have similar immunopathogeneses? Acta Odontol. Scand. 2001;59:235–243. [PubMed]
24. Brown AS, Schaefer CA, Quesenberry CP Jr, Liu L, Babulas VP, Susser ES. Maternal exposure to toxoplasmosis and risk of schizophrenia in adult offspring. Am. J. Psychiatry2005;162:767-73.[PubMed]
25. Buxton D, Innes EA. A commercial vaccine for ovine toxoplasmosis. Parasitology. 1995;110:11–16. [PubMed]
26. Camossi LG, Fornazari F, Richini-Pereira VB, da Silva RC, Cardia DF, Langoni H. Immunization of Wistar female rats with 255-Gy-irradiated Toxoplasma gondii: preventing parasite load and maternofoetal transmission. Exp Parasitol. 2014;145:157-63. [PubMed]
27. Canessa A, Del Bono V, De Leo P, Piersantelli N and Terragna. Cotrimoxazole therapy of Toxoplasma gondii encephalitis in AIDS patients. Eur J Clin Microbiol Inf Dis 1992;11:125-130. [PubMed]
28. Carr A, Tindall B, Brew BJ, Marriott DJ, Harkness JL, Penny R, Cooper DA. Low-dose trimethoprim-sulfamethoxazole prophylaxis for toxoplasmic encephalitis in patients with AIDS. Ann Intern Med 1992;117:106-111. [PubMed]
29. Carral L, Kaufer F, Olejnik P, Freuler C, Durlach R. Prevention of congenital toxoplasmosis in a Buenos Aires hospital. Medicina(B Aires). 2013;73:238-42. [PubMed]
30. Carter CJ. Toxoplasmosis and Polygenic Disease Susceptibility Genes: Extensive Toxoplasma gondii Host/Pathogen Interactome Enrichment in Nine Psychiatric or Neurological Disorders. Carter J Pathog.2013;2013:965046. [PubMed]
31. Centers for Disease Control and Prevention. Parasites - Toxoplasmosis (Toxoplasma infection) 2013, cdc homepage: cdc.gov/parasite/toxoplasmosis.
32. Chahed Bel-Ochi N, Bouratbine A, Mousli M. Enzyme-linked immunosorbent assay using recombinant SAG1 antigen to detect Toxoplasma gondii-specific immunoglobulin G antibodies in human sera and saliva. Clin Vaccine Immunol. 2013;20:468-73. [PubMed]
33. Chang HR, Pechere JC. Effects of roxithromycin on acute toxoplasmosis in mice. Antimicrob Agents Chemother 1987;31 :1147-1149. [PubMed]
34. Chirgwin K, Hafner R, Leport C, et al. Randomized phase II trial of atovaquone with pyrimethamine or sulfadiazine for treatment of toxoplasmic encephalitis in patients with acquired immunodeficiency syndrome: ACTG 237/ANRS 039 Study. AIDS Clinical Trials Group 237/Agence Nationale de Recherche sur le SIDA, Essai 039. Clin Infect Dis 2002;34:1243-50. [PubMed]
35. Cortina-Borja M ,Tan HK,Wallon M, Paul M, Prusa A, Buffolano W. European multi centre study on congenital toxoplasmosis (EMSCOT). Prenatal treatment for serious neurological squeal of congenital toxoplasmosis: an observational prospective cohort study. PLoSMed 2010;7:e1000351. [PubMed]
36. Couvreur J, Desmonts G, Thulliez P. Prophylaxis of congenital toxoplasmosis. Effects of spiramycin on placental infection. J Antimicrob Chemother 1988;22:193-200. [PubMed]
37. Crespo M, Quereda C, Pascual J, Rivera M, Clemente L, Cano T. Patterns of sulfadiazine acute nephrotoxicity. Clin Nephrol 2000;54:68-72. [PubMed]
38. Dando C, Gabriel KE, Remington JS, Parmley SF. Simple and efficient method for measuring anti-Toxoplasma immunoglobulin antibodies in human sera using complement-mediated lysis of transgenic tachyzoites expressing beta-galactosidase. J Clin Microbiol. 2001;39:2122-5. [PubMed]
39. Dannemann B, McCutchan JA, Israelski D, et al Antoniskis D, Leport C, Luft B, Nussbaum J, Clumeck N, Moriat P, Chiu J, Vilde JL, Orellana M, Feigal D, Bartok A, Heseltine P, Leedom J, Remington JS, and The California Collaborative Treatment Group. Treatment of toxoplasmic encephalitis in patients with AIDS: a randomized trial comparing pyrimethamine plus clindamycin to pyrimethamine plus sulfadiazine. Ann Intern Med 1992;116:33-43. [PubMed]
40. De Gans J, Portegies P, Reiss P, Troost D, Van Gool T, Lange JM. Pyrimethamine alone as maintenance therapy for central nervous system toxoplasmosis in 38 patients with AIDS. J Acquir Immune Defic Syndr 1992;5:137-142. [PubMed]
41. Dehkordi FS, Borujeni MR, Rahimi E, Abdizadeh R. Detection of Toxoplasma gondii in raw caprine, ovine, buffalo, bovine, and camel milk using cell cultivation, cat bioassay, capture ELISA, and PCR methods in Iran. Foodborne Pathog Dis 2013;10:120-5. [PubMed]
42. Demar M, Ajzenberg D, Maubon D, Djossou F, Panchoe D, Punwasi W, Valery N, Peneau C, Daigre JL, Aznar C, Cottrelle B, Terzan L, Dardé ML, Carme B. Fatal outbreak of human toxoplasmosis along the Maroni River: epidemiological, clinical, and parasitological aspects. Clin Infect Dis 2007;45:e88-95. [PubMed]
43. Derouin F, Chastang C. Enzyme immunoassay to assess effect of antimicrobial agents on Toxoplasma gondii in tissue culture. Antimicrob Agents Chemother 1988;36:1204-1210. [PubMed]
44. Derouin F, Devergie A, Auber P, Gluckman E, Beauvais B, Garin YJF, Lariviere M. Toxoplasmosis in bone marrow-transplant recipients: report of seven cases and review. Clin.Infect.Dis. 1992;15: 67-270. [PubMed]
45. Derouin F, Jacqz Aigrain E, Thulliez P, Couvreur J, Leport C. Cotrimoxazole for prenatal treatment of congenital toxoplasmosis. Parasitology Today 2000;16:254-256. [PubMed]
46. Derouin F, Leport C, Pueyo S, Morlat P, Letrillart B, Chene G, Ecobichon JL, Luft B, Aubertin J, Hafner R, Vilde JL and ANRS 005/ACTG 154 Trial Group. Predictive value of Toxoplasma gondiiantibody titres on the occurrence of toxoplasmic encephalitis in HIV-infected patients. AIDS 1996;10:1521-1527. [PubMed]
47. Desmonts G, Couvreur J. Congenital toxoplasmosis: a prospective study of 378 pregnancies. N Engl J Med 1974;290:1110-1116. [PubMed]
48. Devleesschauwer B, Ale A, Torgerson P, Praet N, Maertens de Noordhout C, Pandey BD, Pun SB, Lake R, Vercruysse J, Joshi DD, Havelaar AH, Duchateau L, Dorny P, Speybroeck N. The burden of parasitic zoonoses in Nepal: a systematic review. PLoSNegl. Trop. Dis. 2014;8:e2634. [PubMed]
49. di Carlo P, Romano A, Casuccio A, Cillino S, Schimmenti MG, Mancuso G, la Chiusa S, Novara V, Ingrassia D, Li Vecchi V, Trizzino M, Titone L. Investigation and management of Toxoplasma gondii infection in pregnancy and infancy: a prospective study. Acta Pharmacol. Sin.2011;32:1063-70. [PubMed]
50. Djurkovic-Djakovic O, Romand S, Nobre R, Couvreur J, Thulliez P. Serologic rebound after one year long treatment for congenital toxoplasmosis. Pediatr Infect Dis 2000;19:91-83. [PubMed]
51. Dubey JP. Re-examination of resistance of Toxoplasma gondii tachyzoites and bradyzoites to pepsin and trypsin digestion. Parasitology. 1998;116:43–50. [PubMed]
52. Dubey JP, Hill DE, Jones JL, Hightower AW, Kirkland E, Roberts JM, Marcet PL, Lehmann T, Vianna MC, Miska K, Sreekumar C, Kwok OC, Shen SK, Gamble HR. Prevalence of viableToxoplasma gondii in beef, chicken, and pork from retail meat stores in the United States: risk assessment to consumers. J Parasitol 2005;91:1082-93. [PubMed]
53. Dubey JP, Jones JL. Comments on "detection of Toxoplasma gondii in raw caprine, ovine, buffalo, bovine, and camel milk using cell cultivation, cat bioassay, capture ELISA, and PCR methods in Iran". Foodborne Pathog Dis 2014;11:500-1. [PubMed]
54. Dubey JP, Lago EG, Gennari SM, Su C, Jones JL. Toxoplasmosis in humans and animals in Brazil: high prevalence, high burden of disease, and epidemiology. Parasitology 2012;139:1375-424. [PubMed]
55. Dunn D, Wallon M, Peyron F, Petersen E, Peckharm C, Gilbert. R. Mother-to-child transmission of toxoplasmosis: risk estimates for clinical counselling. Lancet 1999;353:1829-1833. [PubMed]
56. Eyles DE, Coleman N. Synergistic effect of sulfadiazine and daraprim against experimental toxoplasmosis in the mouse. Antibiot Chemother 1953;3:483-490. [PubMed]
57. Epple HJ. Therapy- and non-therapy-dependent infectious complications in inflammatory bowel disease. Dig. Dis. 2009;27:555-9. [PubMed]
58. Erridge C, Duncan SH, Bereswill S, Heimesaat MM. The induction of colitis and ileitis in mice is associated with marked increases in intestinal concentrations of stimulants of TLRs 2, 4, and 5. PLoS One. 2010;5:e9125. [PubMed]
59. Fekadu A, Shibre T, Cleare AJ. Toxoplasmosis as a cause for behaviour disorders--overview of evidence and mechanisms. Folia. Parasitol2010;57:105-113. [PubMed]
60. Fernandez-Martin J, Leport C, Morlat P, Meyohas MC, Chauvin JP, Vilde JL. Pyrimethamine- clarithromycin combination for therapy of acute Toxoplasma encephalitis in patients with AIDS. Antimicrob Agents Chemother 1991;35:2049-2052. [PubMed]
61. Figueroa MS, Bou G, Marti-Belda P, Lopez-Velez R, Guerrero A. Diagnostic value of polymerase chain reaction in blood and aqueous humor in immunocompetent patients with ocular toxoplasmosis. Retina 2000;20: 614-619. [PubMed]
62. Flegr J. Influence of latent Toxoplasma infection on human personality, physiology and morphology: pros and cons of the Toxoplasma-human model in studying the manipulation hypothesis. J. Exp. Biol.2013;216(Pt 1):127-33. [PubMed]
63. Foot AB, Garin YJ, Ribaud P, Devergie A, Derouin F, Gluckman E. Prophylaxis of toxoplasmosis infection with pyrimethamine/sulfadoxine (Fansidar) in bone marrow transplant recipients. Bone Marrow Transplantation 1994;14:241-245. [PubMed]
64. Forestier F, Daffos F, Rainaut M, Desnottes JF, Gaschard JC. Suivi therapeutique foeto-maternel de la spiramycine au cours de la grossesse. Arch Franc Pediatr 1987;44:39-544. [PubMed]
65. Foulon W, Naessens A, Ho-Yen D. Prevention of congenital toxoplasmosis. J Perinat Med 2000;28:337-345. [PubMed]
66. Foulon W, Pinon JM, Stray-Pedersen B, Pollak A, Lappalainen M, Decoster A, Villena I, Jenum PA, Hayde M, Naessens A. Prenatal diagnosis of congenital toxoplasmosis: a multicenter evaluation of different diagnostic parameters. Am J Obstet Gynecol 1999;181:843-847. [PubMed]
67. Foulon W, Villena I, Stray-Pedersen B, Decoster A, Lappalainen M, Pinon JM, Jenum PA, Hedman K, Naessens A. Treatment of toxoplasmosis during pregnancy: a multicenter study of impact on fetal transmission and children's sequellae at age 1 year. Am J Obstet Gynecol 1999;180:410-5. [PubMed]
68. Frenkel JK, Dubey JP, Miller NL. Toxoplasma gondii in cats: fecal stages identified as coccidian oocysts. Science. 1970;167:893–6. [PubMed]
69. Frenkel JK, Hitchings GH. Relative reversal by vitamins (p-aminobenzoic, folic and folinic acids) of the effects of sulfadiazine and pyrimethamine on Toxoplasma in mouse and man. Antibiot Chemother 1957;7:630-638. [PubMed]
70. Frenkel JK, Nelson BM, Arias-Stella J. Immunosuppression and toxoplasmic encephalitis: clinical and experimental aspects. Hum Pathol. 1975; 6:97–111. [PubMed]
71. Freyre A, Falcon J, Mendez J, Gonzalez M. Toxoplasma gondii: an improved rat model of congenital infection. Exp.Parasitol. 2008;120:142–146. [PubMed]
72. Fricker-Hidalgo H, Cimon B, Chemla C, Darde ML, Delhaes L, L'ollivier C, Godineau N, Houze S, Paris L, Quinio D, Robert-Gangneux F, Villard O, Villena I, Candolfi E, Pelloux H. Toxoplasmaseroconversion with negative or transient immunoglobulin M in pregnant women: myth or reality? A French multicenter retrospective study. J Clin Microbiol. 2013;51:2103-11. [PubMed]
73. Guidelines for Prevention and Treatment of Opportunistic Infections in HIV-Infected Adults and Adolescents. Recommendations from CDC, the National Institutes of Health, and the HIV Medicine Association of Infectious Diseases Society of America. April 10, 2009/58(RR04);1-198.
74. Garin JP, Pellerat J, Maillard MA, Woehrle R, Hezez. Bases theoriques de la prevention par la spiramycine de la toxoplasmose congenitale chez la femme enceinte. Presse Med 1968;76:2266.
75. Gazzinelli R, Hieny S, Wynn T, Wolf S, Sher A. Interleukin 12 is required for the T-lymphocyte-independent induction of interferon g by an intracellular parasite and induces resistance in T-cell-deficient hosts. Proceeding of the National Academy of Sciences USA 1993;90:6115-6119. [PubMed]
76. Gilbert R, Gras L. European Multicentre Study on Congenital Toxoplasmosis. Effect of timing and type of treatment on the risk of mother to child transmission of Toxoplasma gondii. BJOG 2003;110:112-20. [PubMed]
77. Girard PM, Landman R, Gaudebout C, Olivares R, Saimot AG, Jelazko P, Gaudebout C, Certain A, Boue F, Bouvet E, et al. Dapsone-pyrimethamine compared with aerosolized pentamidine as primary prophylaxis against Pneumocystis carinii pneumonia and toxoplasmosis in HIV infection. N Engl J Med 1993;1514-1520. [PubMed]
78. Grant IH, Gold JW, Rosenblum M, Niedzwiecki D, Armstrong D. Toxoplasma gondii serology in HIV-infected patients: the development of central nervous system toxoplasmosis in AIDS. AIDS 1990;4:519-521. [PubMed]
79. Grossman PL, Remington JS.The effect of trimethoprim plus sulfamethoxazole on Toxoplasma gondii in vitro and in vivo. Am J Trop Med Hyg 1979;28:445-455. [PubMed]
80. Guerina NG, Hsu H-W, Meissner C, Maguire JH, Lyntield R, Stechenberg B, Abroms L, Pasternack MS, Hoff R, Eaton RB, Grady GF and the New England Regional Toxoplasma Working Group. Neonatal serologic screening and early treatment for congenital Toxoplasma gondii infection. N Engl J Med 1994;330:1858-1863. [PubMed]
81. Guex AC, Radziiwill AJ, Bucher H. Discontinuation of secondary prophylaxis for toxoplasmic encephalitis in human immunodeficiency virus infection after immune restoration with highly active antiretroviral therapy. Clin Inf Dis 2000;30:602-603. [PubMed]
82. Guigue N, Menotti J, Hamane S, Derouin F, Garin YJ. Performance of the BioPlex(R) 2200 flow immunoassay (Bio-Rad) in critical cases of serodiagnosis of toxoplasmosis. Clin Vaccine Immunol. 2014;21:496-500. [PubMed]
83. Habib FA. Post-treatment assessment of acute Toxoplasma infection during pregnancy. J.Obstet.Gynaecol. 2008;28:593–595. [PubMed]
84. Hardy WD, Feinberg J, Finkelstein DM, Power ME, He W, Kaczka C, Frame PT, Holmes M, Waskin H, Fass RJ. A controlled trial of trimethoprim-sulfamethoxazole or aerosolized pentamidine for secondary prophylaxis of Pneumocystis carinii pneumonia in patients with the acquired immunodeficiency syndrome. N Engl J Med 1992;327:1842-1848. [PubMed]
85. Hofflin JM, Remington JS. In vivo synergism of roxithromycin (RU 965) and interferon against Toxoplasma gondii. Antimicrob. Agents Chemother 1987;81:346-348. [PubMed]
86. Hoffmann S, Batz MB, Morris JG Jr. Annual cost of illness and quality-adjusted life year losses in the United States due to 14 foodborne pathogens. J Food Prot. 2012;75:1292-302. [PubMed]
87. Hohlfeld P, Daffos F, Costa JM, Thulliez P, Forestier F, Vidaud M. Prenatal diagnosis of congenital toxoplasmosis with a polymerase-chain-reaction test on amniotic fluid. N Engl J Med 1994;331:695-659. [PubMed]
88. Holliman RE, Johnson JD, Adams S, Pepper JR. Toxoplasmosis and heart transplantation. J Heart Lung Transplant 1991;10:608-610. [PubMed]
89. Hou S. Pregnancy in chronic renal insufficiency and end-stage renal disease. Am J Kidney Dis. 1999;33:235-52. [PubMed]
90. Hou S. Pregnancy in renal transplant recipients. Adv Ren Replace Ther. 2003;10:40-7. [PubMed]
91. House PK, Vyas A, Sapolsky R. Predator cat odors activate sexual arousal pathways in brains of Toxoplasma gondii infected rats. PLoS One. 2011;6:e23277. [PubMed]
92. Howe DK, Honore S, Derouin F, Sibley LD. Determination of genotypes of Toxoplasma gondii strains isolated from patients with toxoplasmosis. J Clin Microbiol 1997;35:1411-1414. [PubMed]
93. Hughes WT, Kennedy W, Shenep JL, Flynn PM, Hetherington SV, Fullen G, Lancaster DJ, Stein DS, Palte S, Rosenbaum D, et al. Safety and pharmacokinetics of 566C80, a hydroxynapthtoquinone with anti-Pneumocystis carinii activity: a phase 1 study in human deficiency virus (HIV) infected men. J Infect Dis 1991;164:170-177. [PubMed]
94. Hughes WT, Oz HS. Successful prevention and treatment of babesiosis with atovaquone. J Infect Dis. 1995;172:1042–1046. [PubMed]
95. Hughes WT , Oz HS. Successful prevention & treatment of babesiosis with atovaquone. The Best of Infectious Diseases. G Keusch (Ed). Mosby-Year Book Inc 1997 pp.194-196.
96. Inaba A, Koh H, Nakashima Y, Nishimoto M, Hayashi Y, Okamura H, Inoue A, Nanno S, Nakane T, Shimono T, Nakamae H, Hino M. Cerebral toxoplasmosis after umbilical cord blood transplantation diagnosed by the detection of anti-Toxoplasma specific IgM antibody in cerebrospinal fluid. Rinsho Ketsueki 2014;55:456-60. [PubMed]
97. Israelski DM, Chmiel JS, Poggensee L, Phair JP, Remington JS. Prevalence of Toxoplasma infection in a cohort of homosexual men at risk of AIDS and toxoplasmic encephalitis. J Acquir Immune Defic Syndr 1993;6:414-418. [PubMed]
98. Jacobson MA, Besch CL, Child C, Hafner R, Matts JP, Muth K, Wentworth DN, Neaton JD, Abrams D, Rimland D, Perez G, Grant IH, Saravolatz LD, Brown LS, Deyton L and The Beirn Community Programs for Clincal Research on AIDS. Prophylaxis with pyrimethamine for toxoplasmic encephalitis in patients with advanced HIV disease: results of a randomized trial. J Infect Dis 1994;169:384-394.[PubMed]
99. Jamieson SE, de Roubaix L-A, Cortina-Borja M, Tan HK, Mui EJ, et al. Genetic and epigenetic factors at COL2A1 and ABCA4 Influence clinical outcome in congenital toxoplasmosis. PLoS ONE2008;3:e2285. [PubMed]
100. Jones JL, Bonetti V, Holland GN, Press C, Sanislo SR, Khurana RN, Montoya JG. Ocular toxoplasmosis in the United States: recent and remote infections. Clin Infect Dis. 2015;60:271-3. [PubMed]
101. Jones JL, Kruszon-Moran D, Sanders-Lewis K, Wilson M. Toxoplasma gondii infection in the United States, 1999-2004, decline from the prior decade. Am J Trop Med Hyg. 2007;77:405-10.[PubMed]
102. Jorde UP, Horowitz HW, Wormser GP. Utility of dapsone for prophylaxis of Pneumocystis carinii pneumonia in trimethoprim-sulfamethoxazole-intolerant, HIV-infected individuals. AIDS 1993;7:355-359. [PubMed]
103. Kaňková S, Sulc J, Křivohlavá R, Kuběna A, Flegr J. Slower postnatal motor development in infants of mothers with latent toxoplasmosis during the first 18 months of life. Early Hum. Dev.2012;88:879-84. [PubMed]
104. Katlama C, DeWit S, O'Doherty E, Van Glabeke M, Clumeck N, and the European Network for Treatment of AIDS Toxoplasmosis Study Group. Pyrimethamine-clindamycin vs. pyrimethamine-sulfadiazine as acute and long term therapy for toxoplasmic encephalitis in patients with AIDS. Clin Infect Dis 1996;22:268-275. [PubMed]
105. Klinker H, Langmann P, Richter E. Plasma pyrimethamine concentrations during long-term treatment for cerebral Toxoplasmosis in patients with AIDS. Antimicrob Agents Chemother 1996;40:1623-1627. [PubMed]
106. Knight K. How pernicious parasites turn victims into zombies. J Exp Biol. 2013;216(Pt 1):i-iv. [PubMed]
107. Kovacs JA. Efficacy of atovaquone in treatment of toxoplasmosis in patients with AIDS. The Lancet 1992;340:637-8. [PubMed]
108. Kovacs JA, Allegra J, Beaver J, Boarman D, Lewis M, Parrillo JE, Chabner B, Masur H. Characterization of de novo folate synthesis in Pneumocystis carinii and Toxoplasma gondii: potential for screening therapeutic agents. J Infect Dis 1989;160:312-320. [PubMed]
109. Leport C, Chene G, Morlat P, Luft BJ, Rousseau F, Pueyo S, Hafner R, Miro J, Aubertin J, Salamon R, Vilde JL and ANRS OO5-ACTG 154 Group Members. Pyrimethamine for primary prophylaxis of toxoplasmic encephalitis in patients with human immunodeficiency virus infection: a double-blind, randomized trial. J Infect Dis 1996;173:91-97. [PubMed]
110. Leport C, Franck J, Chene G, Derouin F, Ecobichon JL, Pueyo S, Miro JM, Luft BJ, Morlat P, Dumon H and ANRS 005-ACTG 154 study group. Immunoblot profile as predictor of toxoplasmic encephalitis in patients infected with human immunodeficiency virus. Clin Diagn Lab Immun 2001;8:579-84. [PubMed]
111. Leport C, Raffi F, Matheron S, Katlama C, Regnier B, Saimot AG, Marche C, Vedrenne C, Vilde JL. Treatment of central nervous system toxoplasmosis with pyrimethamine/sulfadiazine combination in 35 patients with the acquired immunodeficiency syndrome.Efficacy of long-term continuous therapy. Am J Med 1988;84:94-100. [PubMed]
112. Leport C, Vilde J, Katlama C, Regnier B, Matheron S, Saimot AG. Failure of spiramycin to prevent neurotoxoplasmosis in immunosuppressed patients (letter). JAMA 1986;255:2289. [PubMed]
113. Liesenfeld O, Press C, Montoya JG, Gill R, Isaac-Renton JL, Hedman K, Remington JS.False-positive results in immunoglobulin M (IgM) Toxoplasma antibody tests and importance of confirmatory testing: the Platelia Toxo IgM test. J Clin Microbiol 1997;35:174-178. [PubMed]
114. Lidar M, Langevitz P, Barzilai O, Ram M, Porat-Katz BS, Bizzaro N, Tonutti E, Maieron R, Chowers Y, Bar-Meir S, Shoenfeld Y. Infectious serologies and autoantibodies in inflammatory bowel disease: insinuations at a true pathogenic role. Ann NY Acad Sci. 2009;1173:640-8. [PubMed]
115. Lige B, Sampels V, Coppens I. Characterization of a second sterol-esterifying enzyme in Toxoplasma highlights the importance of cholesterol storage pathways for the parasite. Mol. Microbiol.2013;87:951-67. [PubMed]
116. Longuet P, Leport C. Clindamycin treatment of toxoplasmosis in immunocompromised patients. Rev Contemp Pharmacother 1992;3:313-320.
117. Lopes WD, Rodriguez JD, Souza FA, dos Santos TR, dos Santos RS, Rosanese WM, Lopes WR, Sakamoto CA, da Costa AJ. Sexual transmission of Toxoplasma gondii in sheep. Vet.Parasitol. 2013;195(1-2):47-56. [PubMed]
118. Lucet JC, Bailly MP, Bedos JP, Wolff M, Gachot B, Vachon F. Septic shock due to toxoplasmosis in patients infected with the human immunodeficiency virus. Chest 1993;104:1054-1058. [PubMed]
119. Luft BJ, Conley F, Remington JS, Laverdiere M, Wagner KF, Levine JF, Craven PC, Strandberg DA, File TM, Rice N, Meunier-Carpentier F. Outbreak of central nervous system toxoplasmosis in Western Europe and North America. Lancet 1983;1:781-783. [PubMed]
120. Luft BJ, Hafner R, Korzun H, Leport C, Antoniskis D, Bosler EM, Bourland DD, Uttamchandani R, Fuhrer J, Jacobson J, Morlat P, Vilde JL, Remington JS and members of the ACTG 077p/ANRS 009 study team. Toxoplasmic encephalitis in patients with acquired immunodeficiency syndrome. N Engl Med 1993;329:995-1000. [PubMed]
121. Luft B, Naot Y, Araujo FG, Stinson EB, Remington JS. Primary and reactivated Toxoplasma infection in patients with cardiac transplants. Ann Intern Med 1983;99:27-31. [PubMed]
122. Mack DG, McLeod R. New micromethod to study the effect of antimicrobial agents on Toxoplasma gondii: comparison of sulfadoxine and sulfadiazine individually and in combination with pyrimethamine and study of clindamycine, metronidazole and cyclosporine A. Antimicrob Agents Chemother 1984;26:26-30. [PubMed]
123. Mack DG, Johnson JJ, Roberts F, Roberts CW, Estes RG, et al. HLA class II genes modify outcome of Toxoplasma gondii infection. Int J Parasitol.1999:29:1351–1358. [PubMed]
124. Mallolas J, Zamora L, Gatell JM, Miro JM, Vernet E, Valls ME, Soriano E, SanMiguel JG. Primary prophylaxis for Pneumocystis carinii pneumonia: a randomized trial comparing cotrimoxazole, aerosolized pentamidine and dapsone plus pyrimethamine. AIDS 1993;7:59-64. [PubMed]
125. Mandell GL, Bennett JE, Dolin R, Eds. Churchill Livingstone Elsevier, Philadelphia, PA.; and de-la-Torre A, Stanford M, Curi A, Jaffe GJ, Gomez-Marin JE. Therapy for ocular toxoplasmosis. Ocul Immunol Inflamm. 2011;19:314-20.
126. Martino R, Bretagne S, Rovira M, Ullmann AJ, Maertens J, Held T, Deconinck E, Cordonnier C. Toxoplasmosis after hematopoietic stem transplantation. Report of a 5-year survey from the Infectious Diseases Working Party of European Group for Blood and Marrow Transplantation. Bone Marrow Transplantation 2000;25:1111-1114. [PubMed]
127. McAuley J, Boyer KM, Patel D, Mets M, Swisher C, Roizen N, Wolters C, Stein L, Stein M, Schey W, et al. Early and longitudinal evaluations of treated infants and children and untreated historical patients with congenital toxoplasmosis: the Chicago collaborative treatment trial. Clin Inf Dis 1994;18:38-72. [PubMed]
128. McCabe RE, Luft BJ, Remington JS. Effect of murine interferon gamma on murine toxoplasmosis. J Infect Dis 1984;150:961-962. [PubMed]
129. McCabe RE, Luft BJ, Remington JS. The effects of cyclosporine on Toxoplasma gondii in vivo and in vitro. Transplantation 1986;41:611-615. [PubMed]
130. McLeod R, Boyer K, Karrison T, Kasza K, Swisher C, et al. Outcome of treatment for congenital toxoplasmosis, 1981–2004: the National Collaborative Chicago-Based, Congenital Toxoplasmosis Study. Clin Infect Dis.2006:42:1383–1394. [PubMed]
131. McLeod R, Mack D, Foss R. Levels of pyrimethamine in sera and cerebrospinal and ventricular fluid from infants treated for congenital toxoplasmosis. Antimicrob Agents Chemother 1992;36:1040-1048. [PubMed]
132. Melo MB, Nguyen QP, Cordeiro C, Hassan MA, Yang N, McKell R, Rosowski EE, Julien L, Butty V, Dardé ML, Ajzenberg D, Fitzgerald K, Young LH, Saeij JP. Transcriptional analysis of murine macrophages infected with different Toxoplasma strains identifies novel regulation of host signaling pathways. PLoS Pathog. 2013;9(12):e1003779. [PubMed]
133. Mets MB, Holfels E, Boyer KM. Eye manifestations of congenital toxoplasmosis. Am J Ophthalmol 1996;122:309-324. [PubMed]
134. Mizgajska-Wiktor H, Jarosz W, Andrzejewska I, Krzykała M, Janowski J, Kozłowska M. Differences in some developmental features between Toxoplasma gondii-seropositive and seronegative school children. Folia Parasitol (Praha). 2013;60:416-24. [PubMed]
135. Moncada PA, Montoya JG. Toxoplasmosis in the fetus and newborn: an update on prevalence, diagnosis and treatment. Expert Rev Anti Infect Ther. 2012;10(7):815-28. [PubMed]
136. Montoya JG. Laboratory diagnosis of Toxoplasma gondii infection and toxoplasmosis. J Infect Dis 2002;185(Suppl 1):S73-82. [PubMed]
137. Montoya JG, Remington JS. Toxoplasmic chorioretinitis in the setting of acute acquired toxoplasmosis. Clin Infect Dis 1996;23:277-282. [PubMed]
138. Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363:1965-1976. [PubMed]
139. Montoya JG, Remington JS. Management of Toxoplasma gondii infection in pregnancy. Clin Infect Dis 2008;47:554-566. [PubMed]
140. Munir A, Zaman M, Eltorky M. Toxoplasma gondii pneumonia in a pancreas transplant patient. South Med. J. 2000;93(6):614-7. [PubMed]
141. Murat JB, Dard C, Fricker Hidalgo H, Dardé ML, Brenier-Pinchart MP, Pelloux H. Comparison of the Vidas system and two recent fully automated assays for diagnosis and follow-up of toxoplasmosis in pregnant women and newborns. Clin Vaccine Immunol. 2013;20:1203-12. [PubMed]
142. Muzzio DO, Soldati R, Rolle L, Zygmunt M, Zenclussen AC, Jensen F. B-1a B Cells Regulate T Cell Differentiation Associated with Pregnancy Disturbances. Front Immunol. 2014; 5:6. [PubMed]
143. Naessens A, Jenum PA, Pollak A, Decoster A, Lappalainen M, Villena I, Lebech M, Stray-Pedersen B, Hayde M, Pinon JM, Petersen E, Foulon W. Diagnosis of congenital toxoplasmosis in the neonatal period: a multicenter evaluation. J Pediat 1999;135:714-719. [PubMed]
144. Niyongabo T, Leport C, Vilde JL. Utilite de l'acide folinique dans les cytopenies induites par les medicaments antiparasitaires au cours du SIDA. Presse Med 1991;34:1677-1681. [PubMed]
145. Nussenblatt RB, Belfort R. Ocular toxoplasmosis. An old disease revisited. JAMA 1994;271:304-307. [PubMed]
146. O'Driscoll JC, Holliman RE. Toxoplasmosis and bone marrow transplantation. Rev Med Microbiol 1991;2:215-222.
147. Olariu TR, Remington JS, Montoya JG. Polymerase Chain Reaction in Cerebrospinal Fluid for the Diagnosis of Congenital Toxoplasmosis. Pediatr Infect Dis J. 2014;33:566-70 [PubMed]
148. Oz HS. Toxoplasmosis Complications and Novel Therapeutic Synergism Combination of Diclazuril plus Atovaquone. Frontiers in Microbiology. 2014;5:484 [PubMed]
149. Oz HS. Novel Synergistic Protective Efficacy of Atovaquone and Diclazuril on Fetal Maternal Toxoplasmosis. International J Clinical Medicine 2014;5(15):921-932. [PubMed]
150. Oz HS. Maternal and congenital toxoplasmosis, currently available and novel therapies in horizon. In Parasites in the tropics-a new paradigm shift Frontier in Microbiology 2014;5:385. [PubMed]
151. Oz HS. Toxoplasmosis, Pancreatitis, Obesity and Drug Discovery. Pancreat Disord Ther. 2014;4(2). pii: 138. [PubMed]
152. Oz HS. Fetal and Maternal Toxoplasmosis, Book Chapter 1 in “Recent Advances in Toxoplasmosis Research” CM Lee (Ed) Nova Science Publication Lexington 2014 pp1-33.
153. Oz HS, Chen T, de Villiers WJS. Green Tea Polyphenols and Sulfasalazine have Parallel Anti-inflammatory Properties in Colitis Models, Front Immunol 2013;4:132:1-10. [PubMed]
154. Oz HS, Chen T, Ebersole JL, A Model for Chronic Mucosal Inflammation in IBD and Periodontitis Dig Dis Sci 2010;55:2194-2002. [PubMed]
155. Oz HS, Hughes WT. Novel anti-pneumocystis carinii effects of the immunosuppressant mycophenolate mofetil in contrast to provocative effects of tacrolimus, sirolimus and dexamethasone. J Infect Dis 1997;175:901-904. [PubMed]
156. Oz HS, Hughes WT. Pneumocystis Carinii infection alters GTP-binding proteins in the lung. J. Parasitol. 1997;83:679-685. [PubMed]
157. Oz HS, Tobin T. Diclazuril Protects Against Maternal Gastrointestinal Syndrome and Congenital Toxoplasmosis. Int J Clinic Med, 2014;5:93-101. [PubMed]
158. Oz HS, Tobin T. Atovaquone Ameliorate Gastrointestinal Toxoplasmosis Complications in a Pregnancy model. Medical Science Monitor, 2012;18:BR337-345. [PubMed]
159. Oz HS, Westlund KH. “Human Babesiosis” an emerging transfusion dilemma. Int J Hepatol. 2012;2012:431761. [PubMed]
160. Patel DV, Holfels EM, Vogel NP, Boyer KM, Mets MB, Swisher CN, Roizen NJ, Stein LK, Stein MA, Hopkins J, Witters SE, Mack DG, Luciano RA, Meier P, Remington JS, McLeod R.. Resolution of intracranial calcifications in infants with treated congenital toxoplasmosis. Radiology 1996;199:433-440. [PubMed]
161. Pearson PA, Piracha AR, Sen HA, Jaffe GJ. Atovaquone for the treatment of Toxoplasma retinochoroiditis in immunocompetent patients. Ophthalmology 1999;10:148-153. [PubMed]
162. Pedersen MG, Mortensen PB, Norgaard-Pedersen B, Postolache TT. Toxoplasma gondii infection and self-directed violence in mothers. Arch Gen Psych2012;69:1123-1130. [PubMed]
163. Pernas L, Ramirez R, Holmes TH, Montoya JG, Boothroyd JC. Immune profiling of pregnant Toxoplasma-infected US and Colombia patients reveals surprising impacts of infection on peripheral blood cytokines. J Infect Dis. 2014;210:923-31. [PubMed]
164. Petersen E. Borobio MV, Guy E, Liesenfeld O, Meroni V, Naessens A, Spranzi E, Thulliez P. European multicenter study of the LIAISON automated diagnostic system for determination ofToxoplasma gondii-specific immunoglobulin G (IgG) and IgM and the IgG avidity index. J Clin Microbiol 2005;43:1570-1574. [PubMed]
165. Petersen E, Schmidt DR. Sulfadiazine and pyrimethamine in the postnatal treatment of congenital toxoplasmosis: what are the options? Expert Rev Anti Infect Ther. 2003;1:175-82. [PubMed]
166. Peyron F, Lobry JR, et al. Serotyping of Toxoplasma gondii in chronically infected pregnant women: predominance of type II in Europe and types I and III in Colombia (South America). Microbes Infect. 2006;8:2333-40. [PubMed]
167. Peyron F, Wallon M, Bernardoux C. Long term follow-up of congenital ocular toxoplasmosis. N Engl J Med. 1996;334: 993-994. [PubMed]
168. Piketty C, Derouin F, Rouveix B, Pocidalo JJ. In vivo assessment of antimicrobial agents against Toxoplasma gondii by quantification of parasites in the blood, lungs and brain of infected mice. Antimicrob. Agents Chemother 1990;34:1467-1472. [PubMed]
169. Podzamczer D, Santin M, Jimenez J, Casanova A, Bolao F, Gudiol GR. Thrice-weekly cotrixomazole is better than weekly dapsone-pyrimethamine for the primary prevention of Pneumocystis carinii in HIV-infected patients. AIDS 1993;7:501-506. [PubMed]
170. Portugal LR, Fernandes LR, Alvarez-Leite JI. Host cholesterol and inflammation as common key regulators of toxoplasmosis and artherosclerosis development. Expert Rev Anti Infect Ther.2009;7:807-19. [PubMed]
171. Public Health Service, Dept. of Health and Human Services (US) and Food and Drug Administration (FDA). FDA Public Health Advisory: Limitations of Toxoplasma IgM Commerical Test Kits. Rockville: Department of Health and Human Services, Food and Drug Administration, Vol. 3, 1997.
172. Remington JS, McLeod R, Thulliez P, Desmonts G. Toxoplasmosis. in Remington JS, Klein JO, eds. Infectious diseases of the fetus and newborn infant. 5th ed. Philadelphia; W.B Saunders, 2001;205-346.
173. Remington JS, Vilde JL. Clindamycin for Toxoplasma encephalitis in AIDS. Lancet 1991;338:1142-1143. [PubMed]
174. Remington JS, McLeod R, Thulliez P, Desmonts G. Toxoplasmosis. In ‘‘Infectious Diseases of the Fetus and Newborn Infant’’ (Remington JS, Klein JO, Wilson CB, Baker CJ, eds), 6th edn, 2006, pp. 947–1091. Philadelphia: Elsevier Saunders.
175. Ribera E, Fernandez-Sola A, Juste C, Rovira A, Romero FJ, Armadans-Gil L, Ruiz I, Ocana I, Pahissa A. Comparison of high and low doses of trimethoprim-sulfamethoxazole for primary prevention of toxoplasmic encephalitis in human immunodeficiency virus-infected patients. Clin Infect Dis 1999;29:1461-1466. [PubMed]
176. Roizen N, Swisher CN, Stein MA, Hopkins J, Boyer KM, Holfels E, Mets MB, Stein L, Pattet D, Meier P, Witthers S, Remington JS, Mack D, Heydemann P, Patton D, McLeod R. Neurologic and developmental outcome in treated congenital toxoplasmosis. Pediatrics 1995;95:11-20. [PubMed]
177. Rolan PE, Mercer AJ,Tate E, Benjamin I, Posner J. Disposition of atovaquone in humans. Antimicrob.Agents Chemother. 1997;41:1319–1321. [PubMed]
178. Romand S, Della Bruna C, Farinotti R, Derouin F. In vitro and in vivo effects of rifabutine alone or combined with atovaquone against Toxoplasma gondii. Antimicrob Agents Chemother 1996;40:2015-2020. [PubMed]
179. Romand S, Pudney M, Derouin F. In vitro and in vivo activities of the hydroxynaphtoloquinolone atovaquone alone or combined with pyrimethamine, sulfadiazine, clarithromycin and minocyclin against Toxoplasma gondii. Antimicrob Agents Chemother 1993;37:2371-2378. [PubMed]
180. Romand S, Wallon M, Franck J, Thulliez P, Peyron F, Dumon H. Prenatal diagnosis using polymerase chain reaction on amniotic fluid for congenital toxoplasmosis. Obstet Gynecol 2001;97: 296-300. [PubMed]
181. Rosso F, Les JT, Agudelo A, Villalobos C, Chaves JA, Tunubala GA, Messa A, Remington JS, Montoya JG. Prevalence of Infection with Toxoplasma gondii among Pregnant Women in Cali, Colombia, South America. Amer J Trop Med Hyg. 2008;78:504-508. [PubMed]
182. Ruskin J, Remington JS. Toxoplasmosis in the compromised host. Ann Intern Med 1976;84:193-199. [PubMed]
183. Sabin AB, Feldman HA. Dyes as microchemical indicators of a new immunity phenomenon affecting a protozoon parasite (Toxoplasma). Science. 1948;108:660–3. [PubMed]
184. Saeij JP, Boyle JP, Boothroyd JC. Differences among the three major strains of Toxoplasma gondii and their specific interactions with the infected host. Trends Parasitol 2005;21:476-81113. [PubMed]
185. Schneider MM, Hoepelman AI, Eeftinck Schattenkerk JK, Nielsen TL, van der Graaf Y, Frissen JP, van der Ende IM, Kolsters AF, Borleffs JC. A controlled trial of aerosolized pentamidine or trimethoprim-sulfamethoxazole as primary prophylaxis against Pneumocystis carinii pneumonia in patients with human immunodeficiency virus infection. N Engl J Med 1992;327:1836-41. [PubMed]
186. Shapira Y, Agmon-Levin N, Selmi C, Petríková J, Barzilai O, Ram M, Bizzaro N, Valentini G, Matucci-Cerinic M, Anaya JM, Katz BS, Shoenfeld Y. Prevalence of anti-Toxoplasma antibodies in patients with autoimmune diseases. J Autoimmun. 2012;39(1-2):112-6. [PubMed]
187. Sharif M, Sarvi S, Shokri A, Hosseini Teshnizi S, Rahimi MT, Mizani A, Ahmadpour E, Daryani A. Toxoplasma gondii infection among sheep and goats in Iran: A systematic review and meta-analysis. Parasitol Res. 2014 Nov 8. [PubMed]
188. Sharma SD, Hofflin JM, Remington JS. In vivo recombinant interleukin 2 administration enhances survival against a lethal challenge with Toxoplasma gondii. J Immunol 1985;135:4160-4163. [PubMed]
189. Sibley LD, Boothroyd JC. Virulent strains of Toxoplasma gondii comprise a single clonal lineage. Nature 1992;359:82-85. [PubMed]
190. Slavin MA, Meyers JD, Remington JS, Hackman RC. Toxoplasma gondii infection in marrow transplant recipients: a 20 years experience. Bone Marrow Transplantation 1994;13:549-557. [PubMed]
191. Solbreux P, Sonnet J and Zech F. A retrospective study about the use of cotrimoxazole as diagnositc support and treatment of suspected cerebral toxoplasmosis in AIDS. Acta Clin Belg 1990;45:85-96. [PubMed]
192. Speirs GE, Hakim TG, Calne RY, Wreghitt TG. Relative risk of donnor acquired Toxoplasma gondii in heart, liver and kidney transplant recipients. Clin Transplant 1988;2:257-260.
193. Srivastava IK, Vaidya AB. A mechanism for the synergistic anti-malarial action of atovaquone and proguanil. Antimicrob.AgentsChemother. 1999;43:1334–1339. [PubMed]
194. Stray-Pederson B and The European Research Network on Congenital Toxoplasmosis. Azithromycin levels in placental tissue, amniotic fluid and blood. In: Program and abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy (New Orleans. Sept. 15-18th 1996) Washington DC: American Society for Microbiology 1996.
195. Sumi M, Aosai F, Norose K, Takeda W, Kirihara T, Sato K, Fujikawa Y, Shimizu I, Ueki T, Hirosima Y, Ueno M, Ichikawa N, Watanabe M, Kobayashi H. Acute exacerbation of Toxoplasma gondiiinfection after hematopoietic stem cell transplantation: five case reports among 279 recipients. Int J Hematol 2013;98:214-22.
196. Suzuki Y, Conley FK, Remington JS. Treatment of toxoplasmic encephalitis in mice with recombinant gamma interferon. Infect Immunol 1990;58:3050-3055. [PubMed]
197. Swisher CN, Boyer K, McLeod R, The Toxoplasmosis Study Group. Congenital toxoplasmosis. Semin Pediatr Neurol 1994;1:4-25. [PubMed]
198. Teixeira LE, Kanunfre KA, Shimokawa PT, Targa LS, Rodrigues JC, Domingues W, Yamamoto L, Okay TS. The performance of four molecular methods for the laboratory diagnosis of congenital toxoplasmosis in amniotic fluid samples. Rev Soc Bras Med Trop 2013;46:584-8. [PubMed]
199. Thiebaut R, Leproust S, Chene G and Gilbert R. Effectiveness of prenatal treatment for congenital toxoplasmosis: a meta-analysis of individual patients' data. Lancet 2007;369:115-22. [PubMed]
200. Thiermann E, Apt W, Atias A, Lorca M, Olguin J. A comparative study of some combined treatment regimens in acute toxoplasmosis in mice. Am J Trop Med Hyg 1978;27:747-750. [PubMed]
201. Torre D, Casari S, Speranza F, Donisi A, Gregis G, Poggio A, Ranieri S, Orani A, Anggarano G, Chiodo F, Fiori G, Carosi G and the Italian Collaborative Study Group. Randomized trial of trimethoprim-sulfamethoxazole versus pyrimethamine-sulfadiazine for therapy of toxoplasmic encephalitis in patients with AIDS. Antimicrob Agents Chemother 1998;42:1346-1349. [PubMed]
202. Torre D, Speranza F, Martegani R, Zeroli C, Banfi M and Airoldi M. A retrospective study of treatment of cerebral toxoplasmosis in AIDS patients with trimethoprim-sulphamethoxazole. J Infect 1998;37:15-18. [PubMed]
203. Torrey EF, Bartko JJ, Lun ZR, Yolken RH. Antibodies to Toxoplasma gondii in patients with schizophrenia: a meta-analysis. Schizophr. Bull2007;33:729-736. [PubMed]
204. USPHS/ISDA Prevention of opportunistic infections working group: USPHS/IDSA guidelines for the prevention of opportunistic infections in persons infected with human immunodeficiency virus. Morbity and Mortality Weekly Report 1999. 2001. [PubMed]
205. Van Delden C, Hirschel B. Folinic acid supplements to pyrimethamine-sulfadiazine for Toxoplasma encephalitis are associated with better outcome. J Infect Dis 1996;173:1294-1295. [PubMed]
206. Vergani P, Ghidini A, Ceruti P, Strobelt N, Spelta A, Zapparoli B, Rescaldani R. Congenital toxoplasmosis: efficacy of maternal treatment with spiramycin alone. Am J Reproduct Immunol 1998;39:335-340. [PubMed]
207. Villena I, Aubert D, Leroux B, Dupouy D, Talmud M, Chemla C, Trenque T, Schmit G, Quereux C, Guenounou M, Pluot M, Bonhomme A, Pinon JM. Pyrimethamine-sulfadoxine treatment of congenital toxoplasmosis: follow-up of 78 cases between 1980 and 1997. Scand J Infect Dis 1998;30:295-300. [PubMed]
208. Wallon M, Liou C, Garner P, Peyron F. Congenital toxoplasmosis: systematic review of evidence of efficacy of treatment in pregnancy. Brit Med J 1999;318:1511-1514. [PubMed]
209. Wanderley FS, Porto WJ, Câmara DR, da Cruz NL, Feitosa BC, Freire RL, de Moraes EP, Mota RA. Experimental vaginal infection of goats with semen contaminated with the "CPG" strain ofToxoplasma gondii. J.Parasitol. 2013;99:610-3. [PubMed]
210. Wanke C, Tuazon CU, Kovacs A, Dina T, Davis DO, Barton N, Katz D, Lunde M, Levy C, Conley FK, et al. Toxoplasma encephalitis in patients with acquired immune deficiency syndrome: diagnosis and response to therapy. Am J Trop Med Hyg 1987;36:509-516. [PubMed]
211. Wang HL, Wang GH, Li QY, Shu C, Jiang MS, Guo Y. Prevalence of Toxoplasma infection in first-episode schizophrenia and comparison between Toxoplasma-seropositive and Toxoplasma-seronegative schizophrenia. Acta Psychiatr Scand 2006;114:40-8. [PubMed]
212. Weiss LM, Harris C, Berger M, Harris C, Berger M. Pyrimethamine concentrations in serum and cerebrospinal fluid during treatment of acute Toxoplasma encephalitis in patients with AIDS. J Infect Dis 1988;157:580-583. [PubMed]
213. Wilson CB, Remington JS, Stagno S, Reynolds DW. Development of children born with subclinical congenital Toxoplasma infection. Pediatrics 1980;66:767-774. [PubMed]
214. Wolf A, Cowen D, Paige B. Human toxoplasmosis: occurrence in infants as an encephalomyelitis verification by transmission to animals. Science1939;89:226–227. [PubMed]
215. Wreghitt TG, Hakim M, Gray JJ, Balfour AH, Stovin PG, Stewart S, Scott J, English TA, Wallwork J. Toxoplasmosis in heart and heart and lung transplant recipients. J Clin Path 1989;42:194-199. [PubMed]
216. Zhang Y, Traskman-Bendz L, Janelidze S, Langenberg P, Saleh A, Constantine N, Okusaga O, Bay-Richter C, Brundin L, Postolache TT. Toxoplasma gondii immunoglobulin G antibodies and nonfatal suicidal self-directed violence. J Clin Psychiatry2012;73:1069-1076. [PubMed]
Tables & Figures
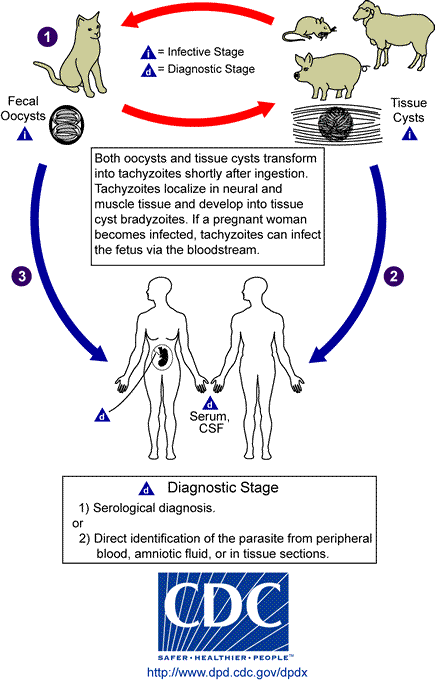
Members of the cat family (Felidae) are the only known definitive hosts for the sexual stages of T. gondii and thus are the main reservoirs of infection. Cats become infected with T. gondii by carnivorism (1). After tissue cysts or oocysts are ingested by the cat, viable organisms are released and invade epithelial cells of the small intestine where they undergo an asexual followed by a sexual cycle and then form oocysts, which are excreted. The unsporulated oocyst takes 1 to 5 days after excretion to sporulate (become infective). Although cats shed oocysts for only 1 to 2 weeks, large numbers may be shed. Oocysts can survive in the environment for several months and are remarkably resistant to disinfectants, freezing, and drying, but are killed by heating to 70°C for 10 minutes.
Human infection may be acquired in several ways: A) ingestion of undercooked infected meat containing Toxoplasma cysts (2); B) ingestion of the oocyst from fecally contaminated hands or food (3); C) organ transplantation or blood transfusion; D) transplacental transmission; E) accidental inoculation of tachyzoites. The parasites form tissue cysts, most commonly in skeletal muscle, myocardium, and brain; these cysts may remain throughout the life of the host.
What's New
Ferguson DJP, et al. Congenital toxoplasmosis: Continued Parasite Proliferation in the Fetal Brain Despite Maternal Immunological Control in Other Tissues. Clin Infect Dis 2013;56:204-208.
Denkers EY, et al. Toxoplasma gondii parasites take control within host cells. Microbe 2012;7:360-365.
Recognizing Eye Infections. Infect Med. 2008:376-384
Sobrin L, et al. Intravitreal clindamycin for toxoplasmic retinochoroiditis. Retina 2007;27:952-957.
Guided Medline Search For:
Reviews
Mege JL, Meghari S, Honstettre A, Capo C, Raoult D. The Two faces of interleukin 10 in human infectious diseases. Lancet Infectious Diseases 2006:7;557-569.
Radke et al. Host Cell-Directed Interactions with Toxoplasma Influence Pathogenesis. Microbe / Volume 2, Number 5, 2007.
Guided Medline Search For Recent Reviews
Guided Medline Search For Historical Aspects
Table of Contents
- Etiology
- Epidemiology
- Clinical Manifestations
- Immunocompetent Patients
- Maternal and Fetal Toxoplasmosis
- Congenital Toxoplasmosis
- Ocular Manifestation of Disease
- Immunosuppressed Patients
- Organ Transplantation, Blood Transfusion and Toxoplasmosis
- Mental Diseases and Toxoplasmosis
- Toxoplasmosis and Autoimmune Disease
- Toxoplasma and Sexual Attraction
- Laboratory and Radiological Diagnostics
- Pathogenesis
- Toxoplasma Genotypes
- Current Available Anti-toxoplasma Therapies
- Therapeutic Indications
- Standard of Care Therapy
- Endpoints for Monitoring Therapy
- Experimental Therapies
- Vaccines
- Preventive Measures
- Prophylactic Therapeutic Agents
