Echinocandins (Anidulafungin, Caspofungin, Micafungin)
Authors: Alexander Imhof, M.D., Kieren A. Marr, M.D.
CLASS
The echinocandins are a class of semisynthetic antifungal lipopeptides that are structurally characterized by a cyclic hexapeptide core linked to variably configured lipid side chains. These drugs inhibit glucan synthase, the enzyme responsible for synthesis of β1-3 linked glucan, a major polysaccharide component of the cell wall of several pathogenic fungi (70). The β 1-3 glucan synthase complex is composed of 2 subunits: a plasma membrane-bound catalytic subunit (FKS) and an activating subunit. The activating subunit, with guanosine triphosphatase activity, activates the catalytic subunit, which polymerizes UDP-glucose into fibrils of glucan and extrudes the polymer through the plasma membrane into the cell wall. Inhibition is thought to be non-competitive, but the precise mechanism by which these drugs interact with the enzyme complex to inhibit synthesis of glucan is not well defined.
Echinocandins have been in development for a number of years, with initial indications targeting infection caused by Pneumocystis and Candida species. Cilofungin was the first echinocandin that reached Phase II clinical development for its activity against Candida sp. (62, 74), but its development was suspended due to toxicites related to the intravenous vehicle (95). Caspofungin acetate was the first echinocandin to receive marketing approval by several countries for the treatment of invasive aspergillosis unresponsive to other therapies and for invasive candidiasis. It is a water-soluble, semisynthetic derivative of pneumocandin B, a fermentation product isolated from the fungus Glarea Zozoyensis. (2) Specifically, it is a l-[(4R,5S)-5-[(2-aminoethyl)amino]-N2-(10,12-dimethyl-1-oxotetradecyl)-4-hydroxy-l-ornithine)-5[(3R)-3-hydroxy-L-ornithine] diacetate salt of pneumocandin B, with the molecular formula C52H88N100152C2H4O2 (Figure 1a) and a molecular weight of 1213.42 d. The other current echinocandins in advanced stages of development are micafungin (Fujisawa Inc, Deerfield, IL, Figure 1b) and anidulafungin (Versicor Inc., Fremont, CA, Figure 1c). These drugs have similar pharmacological properties (54, 70); similarities and differences in drugs within the class will be detailed in following sections. In certain sections, caspofungin is discussed in greater depth compared to the other echinocandins, only because relatively more information has been made available.
ANTIFUNGAL ACTIVITY
Spectrum
All current echinocandins have activity against Candida sp., as well as several molds.
Fungicidal activity against Candida species allow for measurement of clear growth end-points and interpretable Minimal Inhibitory Concentrations (MIC) using reference broth microdilution methods published by the National Committee for Clinical Laboratory Standards (NCCLS) (69). Efficacy of caspofungin, micafungin and anidulafungin have been demonstrated against Candida sp, including azole-resistant strains (9). Minimal inhibitory concentrations of drugs that cause 90% growth inhibition relative to growth controls (MIC90) are summarized in Table 1. Typically, MIC90’s are very low for Candida species, although relatively higher MIC90’s have been noted among several C. parapsilosis and C. guillermondiiisolates (59, 64, 76, 108). Although breakpoints for resistance have not been defined, MIC90’s of Candida species are thought to be well within the susceptible range. Caspofungin also has been demonstrated to have activity against strains of C. albicans and C. parapsilosis growing as biofilms (10, 51, 81).
The relative activity against molds has been particularly difficult to quantify using NCCLS broth dilution methods for conidium-forming filamentous fungi (document M38-A) (68), largely because the drugs cause damage to growing cells comprising filamentous tips and branch points, but do not yield clear endpoints in turbidit using classical broth dilution assays (Figure 2) (15, 59). Minimum inhibitory concentration (MIC) values reported by clinical laboratories also vary greatly with the culture medium used, the incubation time, the assay methodology, and the defined end point of growth inhibition (7, 8, 9, 12, 19, 22, 55). Currently, various experimental methods are being studied for determining the MIC for Aspergillus sp, including examination of morphologic changes of hyphae (Minimal effective concentration, MEC) (31), disk diffusion (8), and E-Test (31). Caspofungin, micafungin and anidulafungin activity reported against various Aspergillus species, as measured using both MIC90 and MECs, are summarized in Table 2.
Studies have also examined the effects of echinocandins on other opportunistic fungi, such as Pneumocystis jiroveci, Histoplasma capsulatum, Coccidioides immitis, andCryptococcus neoformans. Caspofungin and micafungin have variable in vitro activity against H capsulatum, and appear to have a limited treatment role (40, 49, 67). Micafungin exhibited potent activity against the mycelial forms of H. capsulatum, B. dermatitidis, and C. immitis (MIC range, 0.0078 to 0.0625 μg/ml), but it was very weakly active against their yeast-like forms (MIC range, 32 to >64 micro μg/ml). Micafungin was also more active against the mycelial forms than the yeast-like forms of Paracoccidioides brasiliensis, Penicillium marneffei, and S. schenckii (67). Although caspofungin treatment was effective in reducing tissue burdens of C. immitis in mice (38), further in vivo investigation is needed before these drugs can be recommended for treatment of these infections in humans. Echinocandins also have some in vitro activity against Pneumocystis jiroveci. Activity appears to be limited, however, to the cyst form, which has β-(1,3)-glucan in the cell wall; these drugs are not active against the trophozoite which does not rely on β-(1,3)-glucan for structural integrity. Treatment with caspofungin was found to decrease the number of P. jiroveci cysts by 90% in immunosuppressed rats (78). These results suggest a prophylactic role, since the trophozoite is more prevalent in acute disease and the cyst predominates in latent disease, however, without demonstration of definitive activity, these drugs cannot be recommended above other therapeutic alternatives.
As a class, echinocandins have limited activity against Cryptococcus neoformans, (28) which appear to have a target with reduced affinity for the drug, a lesser contribution of β-1,3 linkages to glucan structure, and a reduction of caspofungin activity by melanin (46, 107). However, in vitro studies that examined combinations of drugs against C. neoformansdemonstrated some evidence of synergism between caspofungin and amphotericin B (13). In combination with fluconazole, the addition of caspofungin resulted in synergism and additivity in a strain-dependent manner; no antagonism has been reported (33).
In vitro activity of echinocandins in combination with triazoles and amphotericin products has also been evaluated for Candida species and molds. No evidence of antagonism was noted using the combination of caspofungin and amphotericin B against C. albicans (13). Caspofungin was synergistic with both fluconazole and voriconazole against C. parapsilosis, but indifference was noted in combination with amphotericin B (109). The combination of caspofungin and amphotericin B is synergistic or additive against Aspergillus species in vitro; antagonism has not been reported (8, 13). Experiments with clinical isolates of A. fumigatus found that synergy with caspofungin was observed with either conventional or lipid-complexed amphotericin B (18). Similar results have been reported using the combination of caspofungin and voriconazole (72), and caspofungin with either itraconazole or posaconazole (58, 91). The potential role of echinocandins administered in combination with other antifungal agents for aspergillosis is currently an active area of clinical investigation.
The bulk of in vivo studies of the efficacy of echinocandins for candidiasis have been performed in mice. In immunocompetent and immunosuppressed mouse models, caspofungin was effective against disseminated candidiasis, with its 90% effective dose comparing favorably with that of antifungal agents such as amphotericin B, amphotericin B lipid complex, and fluconazole (1, 2, 13). In addition, caspofungin was noted to decrease tissue-colony counts of C. albicans, C. krusei, and C. glabrata (75, 77). In one study, a single dose of caspofungin was more effective than amphotericin B at clearing C albicans from kidney tissue (1). In another mouse model of invasive candidiasis with C. tropicalis, micafungin more effectively reduced tissue colony forming units compared to fluconazole and amphotericin B (112).
Numerous animal studies have evaluated the efficacy of caspofungin (1, 2, 15) and micafungin (44, 56, 61) for preventing and treating aspergillosis. In general, the echinocandins yielded similar results with amphotericin B with regards to clinical parameters such as survival, but decreasing fungal burden has been difficult to quantify. This may be explained by the drug’s mechanism of action, which could result in fragmented hyphae and a paradoxical increase in colony forming units upon culture.
Anidulafungin has shown activity in animal models of superficial and disseminated candidiasis, P. jiroveci pneumonia, and pulmonary and disseminated aspergillosis (5). It was found to be effective against fluconazole-resistant esophageal candidiasis in immunocompromised rabbits, and prolonged survival in the setting of invasive A. fumigatus infection in neutropenic mice (5, 110).
Pharmacodynamic Effects
Fungicidal Effects
In general, echinocandins have more “cidal” effects against Candida species compared to filamentous organisms. In vitro studies demonstrated concentration-dependent fungicidal activity of caspofungin against several isolates of Candida tropicalis, C. parapsilosis, C. lusitaniae, C. guilliermondii, C. krusei, C. pseudotropicalis, and C. glabrata (12,13, 64). Micafungin showed fungicidal activity against C. albicans, C. glabrata, and C. tropicalis. One study, however, reported that caspofungin lacked fungicidal activity against 11 (46%) of 24 C. guilliermondii isolates (28, 29). In contrast, echinocandins are inhibitory, but not fungicidal against Aspergillus species. Evidence collected using viability dyes indicated that only fungal cells that are actively producing cell wall at hyphal apices and branch points are killed by caspofungin and micafungin (15, 113).
Postantifungal Effects
Echinocandins demonstrate significant postantifungal effects against Candida, but not Aspergillus species. In one study, exposure of C. albicans strains to 1 hour of concentrations above the MIC resulted in a postantifungal effect of >12 hours, measured using time-kill methods. The duration of postantifungal effect was shown to decrease sharply to 0 to 2 hours after 1 hour of exposure to concentrations below the MIC, suggesting that this effect is concentration dependent (28). In contrast, radiometric assays have shown caspofungin to have a relatively short postantifungal effect against A. fumigatus (82). The postantifungal effects of micafungin of Candida sp. was also influenced by the drug concentration, with the highest concentration resulting in the longest observed postantifungal effects. The highest concentration tested, four times the MIC, resulted in a postantifungal effects of more than 9.8 h for 50% of the tested isolates (range, 0.9 to ≥20.1 h) (29).
Effects on Host Immunity
Reports on the effects of human sera on the antifungal activity of caspofungin against Candida and Aspergillus spp are conflicting. In an in vitro preclinical evaluation of caspofungin, the susceptibility of C. albicans to caspofungin was not significantly influenced by the addition of human serum (13); however, addition of 50% mouse serum resulted in a insignificant increase in MIC (decreased susceptibility) (13). However, in another study, human serum potentiated the antifungal activity of caspofungin against Aspergillus (19). Although the exact mechanism of this enhancement is unknown, it does not appear to be related to the presence of complement or formation of an Aspergillus antibody in human sera. The same investigators also examined the effect of human monocytes and macrophages on the antifungal activity of caspofungin against A. fumigatus. Caspofungin cocultured with human monocytes for 24 hours had significantly greater inhibitory activity against Aspergillus hyphal growth compared with controls. Similar results were observed for caspofungin cocultured with human macrophages, but not with polymorphonuclear neutrophils (20). It has been hypothesized that these indirect effects on host defense against Aspergillus species is one possible explanation for the better antifungal activity against A. fumigatus measured in in vivo studies compared to in vitro studies, however more studies are necessary to make definitive conclusions.
MECHANISMS OF ACTION
Echinocandins are noncompetitive inhibitors of the enzyme β-(1,3)-glucan synthase, which catalyzes the polymerization of uridine diphosphate-glucose (UDP-glucose) into β-( 1,3)-glucan, a structural component of the fungal wall. Decreased β-( 1,3)-glucan results in a loss of cell integrity and rigidity (59), and ballooning of cellular contents from the weakened cell wall, which can result in cell lysis.
Glucan synthase is a UDP-glucosyl-transferase located in the fungal cell membrane. Glucan synthesis occurs on the cytoplasmic side of the membrane, and glucan chains get extruded toward the periplasmic space for incorporation into the cell wall (92). In a large number of fungi, the β-( 1,3)-glucan synthase complex is composed of 2 subunits: a plasma membrane-bound catalytic subunit (FKS) and an activating subunit (RHO) (14, 53, 54, 80). The activating subunit, with guanosine triphosphatase activity, activates the catalytic subunit, which then polymerizes UDP-glucose into fibrils of glucan and extrudes the polymer through the plasma membrane (52, 54). Precisely how echinocandins inhibit the function of the β-( 1,3)-glucan synthase enzyme is not currently known, although activity is thought to be through non-competitive mechanisms (16, 59). Cryptococcus neoformans contains only a single copy of a homologue of FKS1 whose expression is essential for cell viability (98). The FKS homologue of C. neoformans appears to be under the transcriptional control of calcineurin (25). It was observed that exposure to caspofungin was associated with a reduction in β-1,6 glucan of the cryptococcal cell wall, raising the possibility of an action in addition to inhibition of β-1,3 glucan synthesis in C. neoformans (32).
MECHANISMS OF RESISTANCE
Organisms Commonly Resistant
Organisms that do not rely on β-(1-3) linked glucan for cell wall integrity, such as Cryptococcus neoformans, are typically less susceptible to echinocandins compared to organisms that contain a large amount of glucan in β-(1-3) linkage. Echinocandins also have little to no in vitro activity against Blastomyces dermatitidis, Fusarium sp., Sporothrix schenckii,Rhizopus sp and other Zygomycetes, and Histoplasma capsulatum. Certain Candida species, such as C. parapsilosis typically demonstrate higher MICs compared to other Candida species, however, the clinical signficance of this observation is uncertain (5, 24, 26, 30, 32, 34, 35, 45, 49, 67).
Mechanisms of Resistance
Resistance to caspofungin has not yet been described during the limited clinical experience to date. However, after C. albicans was repeatedly exposed to subinhibitory concentrations of caspofungin in vitro, a small (1-fold) increase in MIC (from 0.06 to 0.125 μg/mL) was observed (13). Mutations in FKS1 and FKS2 genes were associated with high-levels of resistance (>l0-fold increase in MIC) to echinocandins (52), and deletion of the GNSI gene were associated with low-levels of resistance (<l0-fold increase in MIC) (52). Overexpression of Sbe2p, a Golgi protein involved in cell wall formation, also resulted in a low-level of echinocandins (70a). Development of in vitro resistance in Aspergillus sp. or other pathogenic fungi to echinocandins has not yet been demonstrated.
While efflux pumps are typically an important mechanism associated with resistance of azole antifungals in Candida species and Aspergillus species (60, 85, 86, 87, 93), their role in conferring resistance to echincandins is not currently understood. Early studies reported that echinocandins did not appear to be substrates for efflux pumps (9, 52), but recently published studies reported that constitutive overexpression of the Candida albicans ATP binding cassette transporter, Cdr2p, conferred resistance to caspofungin (88). Whether this is the result of efflux of the drug, or other changes in the cell wall or membrane is currently unknown.
Methods to Overcome or Prevent Resistance
Since microbial resistance to echinocandins has not yet become a large clinical problem, few efforts have been placed into describing preventative mechanisms. However, one older study demonstrated that mutations in the calcineurin-signaling pathway were associated with a hypersensitivity to echinocandins (71). Whether administration of calcineurin inhibitors can decrease resistance, as has been described with azole-resistant Candida isolates, is currently unknown.
PHARMACOKINETICS
Absorption
All echinocandins have a low oral bioavailability and are currently available only by intravenous infusion. Preclinical animal studies showed that caspofungin is minimally absorbed after oral administration, with an absolute bioavailability of only 0.3% to 1% in mice and 9% in dogs (70). Similar studies for anidulafungin demonstrated a fasting absorption of 5 to 10% in dogs (5, 70). It is not likely that oral formulations will become available for any of the current echinocandins.
Distribution
All echinocandins demonstrate a high amount (>95%) of protein binding and relatively low volume of distributions. The volume of distribution for caspofungin estimates 9.67 L at steady state (5, 70, 95). Animal studies have demonstrated extensive tissue distribution for all echinocandins, although these drugs do not cross intact blood-brain barriers and concentrations in brain tissues are relatively low. When given intraperitoneally to mice, caspofungin was distributed into tissues with a volume of distribution estimating 0.1-0.27 L/kg, and the highest drug concentrations were measured in the liver. Tissue:plasma ratios measured were 16 in liver, 2.9 in kidneys, 2.0 in large intestine, 1.3 in small intestine, 1.1 in lungs, 1.0 in spleen, 0.3 in heart, 0.2 in thigh, and 0.1 in brain (43).
Routes of Elimination
Metabolism
All of the echinocandins appear to be metabolized in the liver via mechanisms other than the cytochrome P450 enzyme system (39). Caspofungin is slowly metabolized in the liver by hydrolysis and N-acetylation, with some of the drug excreted relatively unchanged in bile (16, 96)
Renal Excretion
None of the echinocandins are excreted by the kidneys to a great degree. Only 1.44% of the total dose of caspofungin, and 15% of micafungin are excreted unchanged by the kidneys, with total renal clearances of 1 μl/min, and 11 mL/hr/kg, respectively (5).
Pharmacokinetic Parameters
The pharmacokinetic parameters of echinocandins (in adults) are summarized in Table 3. Plasma caspofungin elimination is typically triphasic, with mean α, β, and γ half-lives of 1 to 2, 9 to 11, and 40 to 50 hours, respectively (96). Caspofungin is administered as a single loading dose of 70 mg, followed by 50 mg daily. In normal volunteer studies, a single 70 mg dose of intravenous caspofungin resulted in mean peak plasma concentrations of 10.45 μg/ml and trough concentrations of 1.19 μg/ml. Mean trough concentrations were >1 μg/mL in subjects who received the loading dose, whereas they were <I μg/mL in subjects who did not receive the loading dose. In healthy subjects, multiple dosing at 70 mg IV was associated with moderate accumulation (25%-50% after 3 weeks), with mean trough concentrations of 1.34, 2.43, and 2.64 μg/ml on days 1, 14, and 21 of administration. Excretion of caspofungin and its metabolites is slow, with 75.1% of radioactive products recovered (40.7% in urine, 34.4% in feces).
In a 14-day, parallel-panel, Phase I study, Stone et al., examined the effect of gender on the multiple-dose pharmacokinetics of caspofungin in healthy subjects. Eight men and eight women received a loading dose (70 mg) of caspofungin, followed by 50 mg for 13 days. Plasma concentrations of caspofungin were similar in the men and women on day 1. Based on AUC values on day 14, the women had exposure to a mean 22% more drug (90% CI, 1 to 47). Other pharmacokinetic parameters such as elimination half-life and volume of distribution were not reported. Despite the small difference, no dose adjustment has been recommended based on gender (95). Similarly, although healthy volunteer studies demonstrated that elderly subjects had a 28% mean increase in AUC compared with control subjects, no dose change is recommended based on older age (95). A comparison of the pharmacokinetic characteristics of micafungin in elderly (aged 66–78 y) and non-elderly (aged 20–24 y) patients revealed no significant pharmacokinetic differences between the two groups (89). Finally, there appear to be no clinically significant differences in the pharmacokinetic parameters of caspofungin based on race (16).
The plasma elimination half-life of micafungin is 14 to 15 hours, although a slightly shorter half-life has been reported in adults receiving bone marrow or peripheral stem cell transplantation (10.7 to 13.9 hours). After 2-hour infusions of 25 mg daily in healthy subjects, mean AUC-24 values were 19, 28, and 30 μg x mL/hr on days 1, 4, and 7, respectively. The elimination half life varied in febrile neutropenic pediatric patients, from 12 to 13 hours on day 1 to 21 hours after 4 days of infusion (34). The pharmacokinetics of micafungin were not significantly altered in patients with severe renal dysfunction (GFR less than 30 ml/min)(39).
After oral doses of anidulafungin (up to 1000 mg), an elimination half-life of about 30 hours has been reported in healthy subjects (21). Half-life data following intravenous use in healthy adults revealed 29 hours, and limited data suggest no significant effect of renal impairment on pharmacokinetics (102).
CNS/CSF Distribution
In animal models, echinocandins appear to reach brain tissue after IV infusion, but do not cross into the cerebrospinal fluid to a significant degree. The following brain:plasma ratios have been reported for the different echinocandins: 0.1 for caspofungin, 0.01 for micafungin, and 0.14 for anidulafungin (41, 42, 43). When micafungin was given at 0.5, 1, and 2 mg/kg of body weight intravenously once daily for a total of 8 days in rabbits, the drug was not detectable in cerebrospinal fluid. However, the concentration in brain tissue ranged from 0.08 to 0.18 μg/g (41).
In a pharmacokinetic study of anidulafungin in rabbits, measurable concentrations of anidulafungin in brain tissue were noted at dosages of 0.5 mg/kg in another animal model (42).
Effect of Disease States
Renal Insufficiency
After administration of a single 70-mg IV dose of caspofungin, volunteers with moderate renal insufficiency (creatinine clearance 31-49 mL/min), severe renal insufficiency (5-30 ml/min), and endstage renal disease (<10 mL/min and dialysis dependent) had moderate increases in caspofungin plasma concentrations compared with control subjects, with an increase in AUC ranging from 30% to 49% (16). Nevertheless, based on the finding that mild to severe renal impairment had no remarkable effect on trough concentrations in patients receiving multiple doses of caspofungin (50 mg/d) in clinical trials, the manufacturer does not recommend dose adjustment in patients with renal insufficiency (95).
Micafungin pharmacokinetic parameters were compared between 2 groups of subjects with either normal renal function (creatinine clearance >80 mL/min) or severe renal dysfunction (creatinine clearance <30 mL/min) following single-dose of 100 mg micafungin. Regression analyses failed to demonstrate a significant correlation between creatinine clearance and any of the pharmacokinetic parameters studied. Dose adjustments do not appear to be required in patients with renal impairment (103). Similarly, there was no apparent change in pharmacokinetics of anidulafungin in people who had variable degrees of renal insufficiency (101, 102).
Hepatic Insufficiency
The effect of hepatic insufficiency on caspofungin pharmacokinetics was evaluated in a Phase I pilot study. Eight subjects with mild hepatic insufficiency (Child-Pugh score 5-6) and 8 subjects with moderate hepatic insufficiency (Child-Pugh score 7-9) were administered a single dose of caspofungin (70 mg IV). The extent of absorption of caspofungin (denoted by AUC) increased by 55% (90% CI, 32 to 86) and 76% (90% CI, 51 to 106) in patients with mild and moderate hepatic insufficiency, respectively, compared with historical healthy control subjects (96). Another study evaluated the impact of dose-adjustment of caspofungin on pharmacokinetics in 8 subjects with mild hepatic insufficiency, 8 subjects with moderate hepatic insufficiency, and 16 healthy subjects matched by age, sex, and body weight. Control subjects and those with mild hepatic insufficiency received a 70-mg IV loading dose of caspofungin on day 1 followed by 50 mg IV daily for the remainder (13 days) of the study. Subjects with moderate hepatic insufficiency received a 70-mg loading dose of caspofungin IV on day 1, followed by 35 mg IV daily on days 2 through 14. The AUC on day 14 increased by 21% (90% CI, 4 to 39) and 7% (90% CI,-10 to 28) in subjects with mild and moderate hepatic insufficiency, respectively, compared with control subjects. On the basis of this preliminary study, the investigators did not recommend dose adjustment for patients with mild hepatic insufficiency. In patients with moderate hepatic insufficiency, however, they recommended that after the initial 70-mg loading dose, the maintenance dose be reduced to 35 mg/d. No pharmacokinetic data on caspofungin are available in patients with severe hepatic insufficiency (Child-Pugh score >9) (95). However, in patients with severe hepatic insufficiency (Child-Pugh score>9) treated with anidulafungin 50 mg daily, plasma concentration of anidulafungin is decreased, plasma clearance is increased, and total distribution volume is doubled compared to healthy volunteers (99).
Subjects with moderate hepatic disease (Child–Pugh score 7–9) who received a single 100 mg dose of micafungin exhibited reduction in peak concentration and AUC of approximately 22% compared to healthy subjects. The observed increased clearance was believed to be the result of decreased albumin binding by micafungin and subsequent greater hepatic extraction. Despite the reduced systemic exposure, blood concentrations of micafungin remained above those thought to be necessary for antifungal activity. The authors concluded that among patients with moderate hepatic dysfunction, no dose adjustments would be required (104).
DOSAGE
Adults and Children
The recommended adult dosage for caspofungin is 70 mg IV on day 1 followed by 50 mg/d thereafter. Caspofungin should be infused slowly over 1 hour and is not compatible with any diluent that contains dextrose (16). The duration of treatment depends on the severity of underlying disease, recovery from immunosuppression, and clinical response (17). A single daily dose of 35 mg IV has been recommended for anidulafungin, and micafungin has been administered in doses ranging from 50 mg IV to 150 mg IV (5, 23, 34, 36). In safety studies involving patients undergoing hematopoietic stem cell transplantation, a maximum tolerated dose was not reached for micafungin, despite dose escalation up to 8 mg/kg daily (79).
Micafungin has been administered to children > 2 years of age at a dose of 50 mg (106). Specific recommendations for dosing of caspofungin or anidulafungin in children have not yet been made available, but is a subject of current clinical investigation.
Renal Failure
Although there was indication that caspofungin exposure accumulates in the setting of renal impairment, no dose adjustment has been deemed necessary, based on small differences in total exposure in healthy subject studies and no differences in trough concentrations in phase III treatment studies. Caspofungin is not removed by hemodialysis, so supplemental doses are not required. Likewise, dose adjustment does not appear to be required for micafungin or anidulafungin administration in the setting of moderate to severe renal disease (101, 102).
Hepatic Failure
Dose adjustment for caspofungin was not deemed necessary in the setting of mild hepatic dysfunction, however, in patients with moderate hepatic insuffciency (Child-Pugh 7 to 9), daily maintenance doses should be reduced from 50 mg to 35 mg (after administration of a 70 mg loading dose on the first day) (16). Fewer data exist for micafungin and anidulafungin, but one study showed that dose adjustment for micafungin may not be indicated in the setting of moderate liver disease (103). As only few pharmacokinetic data for echinocandins are available in patients with severe hepatic insufficiency (99), and clinical experience has been limited, use of these drugs is not currently recommended in these patients.
Pregnancy
Caspofungin has been shown to be embryotoxic in animal studies, producing incomplete ossification of the skull and torso and an increased incidence of cervical rib formation. Consequently, caspofungin is designated a Pregnancy Category C drug (animal studies have shown an adverse effect on the fetus, but there are no adequate studies in humans) (16). Although the distribution of caspofungin and its metabolites in human milk is not known, the drug is excreted in the milk of animals at concentrations similar to those in maternal plasma. The use of caspofungin (possibly all echinocandins) in pregnant women and nursing mothers should be avoided.
advERSE EFFECTS
Overall, adverse events (AE) related to echinocandins are generally infrequent and mild. In normal volunteer and clinical studies of caspofungin, the most frequently reported drug-related AEs were mild to moderate infusion-related reactions and headache. Dermatologic effects (eg, flushing, erythema, wheals, or rash), facial edema, or respiratory symptoms (wheezing or bronchoconstriction) associated with histamine release were also noted. The most common laboratory adverse event has been elevation of liver transaminase levels, which have been noted to be dose related, but mild and reversible upon cessation of the drug (16, 95). Eosinophilia was also reported (95).
Adverse events associated with micafungin administration are infrequent and most often mild. The most frequently reported drug-related side effects included nausea, vomiting, bilirubinemia, and increased liver function test levels, none of which were considered serious or dose-limiting (89).
The most common clinical AEs associated with administration of anidulafungin include headache, elevated alanine transaminase (ALT), dizziness and nausea. Occasional infusion reactions have also been observed upon administration of intravenous anidulafungin, which typically include symptoms of nausea, dyspnea, and facial flushing; These symptoms were shown to be unrelated to blood histamine levels (4).
MONITORING REQUIREMENTS
Routine monitoring of blood or tissue levels of echinocandins is not currently recommended. Because of potential complications associated with therapy in these typically complex patients, obtaining routine serum chemistries, hepatic enzymes, and complete blood counts is prudent.
DRUG INTERACTIONS
In general, echinocandins are poor substrates for CYP450 enzymes, and do not interact with P-glycoprotein, which mediates efflux of a variety of drugs from cells (11). Thus, fewer drug interactions have been noted with this class of compounds compared to other antifungals, such as triazoles, which may be both substrates and inhibitors of several CYP450 isoenzymes. One clinically meaningful drug interaction that has been noted is with cyclosporin, although the precise mechanism of this interaction is not completely understood. In healthy subject studies, concurrent cyclosporin administration increased the AUC of caspofungin by approximately 35%, which was associated with mild, reversible elevations in hepatic transaminases. Although administration of caspofungin with cyclosporin has not been recommended because of this potential interaction, clinical experience has accumulated in this setting over the last two years, and current studies are evaluating the clinical significance of co-administration in more depth. Cyclosporin also increases the AUC of micafungin and anidulafungin, but to a lesser degree (13% (105) and 21% (100), respectively); these drugs have been co-administered without dose adjustments in both healthy volunteer studies and clinical studies without apparent complications. None of the echinocandins appear to interact with other calcineurin inhibitors, such as tacrolimus or sirolimus.
Results of regression analyses of pharmacokinetic data from an unspecified small number of patients suggest that concentrations of caspofungin may be reduced by co-administration of inducers or mixed inducer/inhibitors of CYP450 isozymes, such as efavirenz, nelfinavir, nevirapine, phenytoin, rifampin, dexamethasone, and carbamazepine. Although co-administration is not contraindicated, monitoring of caspofungin levels, or dose escalation may be considered in patients who are not responding clinically while concurrently receiving these medications (16).
CLINICAL INDICATIONS
Caspofungin was licensed by the US Food and Drug Administration (FDA) in January 2001 for therapy of invasive aspergillosis in patients who are intolerant to other licensed therapies (amphotericin B, lipid amphotericin B formulations, and/or itraconazole), or have refractory disease (16, 17). To date, multiple countries have licensed caspofungin for similar indications, with more recent licensure obtained for treatment of candidiasis.
The data supporting use of caspofungin for “salvage” therapy of aspergillosis were generated by a small, historic-controlled trial that demonstrated a response rate approximating 40% in patients who received caspofungin after failure (intolerance or refractoriness) of other antifungals (57). This response was considered to be improved compared to that of historic control patients who continued to receive other licensed antifungal products, such as amphotericin formulations. Recently, more attention has been placed into the potential application of echinocandins administered in combination with other antifungals for invasive aspergillosis. Animal models, case reports and small series of patients suggest that caspofungin, administered with triazoles or amphotericin formulations, may result in optimal clinical outcomes (3, 50, 73, 84), however, randomized trials will be necessary to define the utility of echinocandin-based combination therapies.
Randomized trials have also established clinical utility of caspofungin for treatment of oropharyngeal and esophageal candidiasis in adults (6, 37, 47, 111), and invasive candidiasis(66, 94). In a randomized comparative study, caspofungin (70 mg loading dose then 50 mg/d) was equivalent to, but better tolerated than amphotericin B deoxycholate (0.6-1.0 mg/kg/d) for invasive candidiasis (66). Similarly, micafungin and anidulafungin have been evaluated for diseases caused by Candida species, and micafungin has been studied for short-term antifungal prophylaxis in patients undergoing hematopoietic stem cell transplantation. Neither of these drugs have been licensed for clinical use at this time.
CONCLUSION
In summary, echinocandins represent a new class of antifungals that have a novel mechanism of action, targeting cell wall synthesis rather than cell membrane integrity. Three compounds, caspofungin, micafungin, and anidulafungin, have similar activity profiles, with minor differences relating to pharmacokinetic parameters. All of these drugs, which are in various stages of clinical development, should add greatly to the armamentarium of antifungals available for therapy of diseases caused by the most common yeasts and molds.
REFERENCES
1. Abruzzo GK, Flattery AM, Gill CJ, Kong L, Smith JG, Pikounis VB, Balkovec JM, Bouffard AF, Dropinski JF, Rosen H, Kropp H, Bartizal K. Evaluation of the echinocandin antifungal MK-0991 (L-743,872): efficacies in mouse models of disseminated aspergillosis, candidiasis, and cryptococcosis. Antimicrob Agents Chemother 1997;41:2333-8.[PubMed]
2. Abruzzo GK, Gill CJ, Flattery AM, Kong L, Leighton C, Smith JG, Pikounis VB, Bartizal K, Rosen H. Efficacy of the echinocandin caspofungin against disseminated aspergillosis and candidiasis in cyclophosphamide-induced immunosuppressed mice. Antimicrob Agents Chemother 2000;44:2310-8. [PubMed]
3. Aliff TB, Maslak PG, Jurcic JG, Heaney ML, Cathcart KN, Sepkowitz KA, Weiss MA. Refractory Aspergillus pneumonia in patients with acute leukemia: successful therapy with combination caspofungin and liposomal amphotericin. Cancer 2003;97:1025-32. [PubMed]
4. Anidulafungin. Anidulafungin (VER002) Investigator`s Brochure. Edition 009.; October 22, 2001.
5. Arathoon EG. Clinical efficacy of echinocandin antifungals. Curr Opin Infect Dis 2001;14:685-91. [PubMed]
6. Arathoon EG, Gotuzzo E, Noriega LM, Berman RS, DiNubile MJ, Sable CA. Randomized, double-blind, multicenter study of caspofungin versus amphotericin B for treatment of oropharyngeal and esophageal candidiases. Antimicrob Agents Chemother 2002;46:451-7. [PubMed]
7. Arevalo MP, Carrillo-Munoz AJ, Salgado J, Cardenes D, Brio S, Quindos G, Espinel-Ingroff A. Antifungal activity of the echinocandin anidulafungin (VER002, LY-303366) against yeast pathogens: a comparative study with M27-A microdilution method. J Antimicrob Chemother 2003;51:163-6. [PubMed]
8. Arikan S, Paetznick V, Rex JH. Comparative evaluation of disk diffusion with microdilution assay in susceptibility testing of caspofungin against Aspergillus and Fusarium isolates. Antimicrob Agents Chemother 2002;46:3084-7.[PubMed]
9. Bachmann SP, Patterson TF, Lopez-Ribot JL. In vitro activity of caspofungin (MK-0991) against Candida albicans clinical isolates displaying different mechanisms of azole resistance. J Clin Microbiol 2002;40:2228-30. [PubMed]
10. Bachmann SP, VandeWalle K, Ramage G, Patterson TF, Wickes BL, Graybill JR, Lopez-Ribot JL. In vitro activity of caspofungin against Candida albicans biofilms. Antimicrob Agents Chemother 2002;46:3591-6. [PubMed]
11. Balani SK, Xu X, Arison BH, Silva MV, Gries A, DeLuna FA, Cui D, Kari PH, Ly T, Hop CE, Singh R, Wallace MA, Dean DC, Lin JH, Pearson PG, Baillie TA. Metabolites of caspofungin acetate, a potent antifungal agent, in human plasma and urine. Drug Metab Dispos 2000;28:1274-8. [PubMed]
12. Barchiesi F, Schimizzi AM, Fothergill AW, Scalise G, Rinaldi MG. In vitro activity of the new echinocandin antifungal, MK-0991, against common and uncommon clinical isolates of Candida species. Eur J Clin Microbiol Infect Dis 1999;18:302-4.[PubMed]
13. Bartizal K, Gill CJ, Abruzzo GK, Flattery AM, Kong L, Scott PM, Smith JG, Leighton CE, Bouffard A, Dropinski JF, Balkovec J. In vitro preclinical evaluation studies with the echinocandin antifungal MK-0991 (L-743,872). Antimicrob Agents Chemother 1997;41:2326-32.[PubMed]
14. Beauvais A, Bruneau JM, Mol PC, Buitrago MJ, Legrand R, Latge JP. Glucan synthase complex of Aspergillus fumigatus. J Bacteriol 2001;183:2273-9. [PubMed]
15. Bowman JC, Hicks PS, Kurtz MB, Rosen H, Schmatz DM, Liberator PA, Douglas CM. The antifungal echinocandin caspofungin acetate kills growing cells of Aspergillus fumigatus in vitro. Antimicrob Agents Chemother 2002;46:3001-12. [PubMed]
16. Cancidas. In. Whithouse Station, NJ: Merck&Co, Inc; 2001.
17. Cancidas (Caspofungin for intravenous injection) Merck Corporation NBdfADPACm, 10 January 2001.
18. Chandrasekar PH, Cutright JL, Manavathu EK. In vitro activity of amphotericin B lipid complex in 2-drug combination with caspofungin against aspergillus fumigatus. In: Focus on fungal infections 12; 2001; Phoenix: Alpharetta, GA: Imedex; 2001.
19. Chiller T, Farrokhshad K, Brummer E, Stevens DA. Influence of human sera on the in vitro activity of the echinocandin caspofungin (MK-0991) against Aspergillus fumigatus. Antimicrob Agents Chemother 2000;44:3302-5.[PubMed]
20. Chiller T, Farrokhshad K, Brummer E, Stevens DA. The interaction of human monocytes, monocyte-derived macrophages, and polymorphonuclear neutrophils with caspofungin (MK-0991), an echinocandin, for antifungal activity against Aspergillus fumigatus. Diagn Microbiol Infect Dis 2001;39:99-103.[PubMed]
21. Chiou CC, Groll AH, Walsh TJ. New drugs and novel targets for treatment of invasive fungal infections in patients with cancer. Oncologist 2000;5:120-35. [PubMed]
22. Chryssanthou E, Cuenca-Estrella M. Comparison of the Antifungal Susceptibility Testing Subcommittee of the European Committee on Antibiotic Susceptibility Testing proposed standard and the E-test with the NCCLS broth microdilution method for voriconazole and caspofungin susceptibility testing of yeast species. J Clin Microbiol 2002;40:3841-4.[PubMed]
23. Cornely OA, Schmitz K, Aisenbrey S. The first echinocandin: caspofungin. Mycoses 2002;45 Suppl 3:56-60.[PubMed]
24. Serrano Mdel C, Valverde-Conde A, Chavez M M, Bernal S, Claro RM, Peman J, Ramirez M, Martin-Mazuelos E. In vitro activity of voriconazole, itraconazole, caspofungin, anidulafungin (VER002, LY303366) and amphotericin B against aspergillus spp. Diagn Microbiol Infect Dis 2003;45:131-5. [PubMed]
25. Del Poeta M, Cruz MC, Cardenas ME, Perfect JR, Heitman J. Synergistic antifungal activities of bafilomycin A(1), fluconazole, and the pneumocandin MK-0991/caspofungin acetate (L-743,873) with calcineurin inhibitors FK506 and L-685,818 against Cryptococcus neoformans. Antimicrob Agents Chemother 2000;44:739-46. [PubMed]
26. Denning DW. Echinocandins and pneumocandins--a new antifungal class with a novel mode of action. J Antimicrob Chemother 1997;40:611-4. [PubMed]
27. Eli. LY303366 Clinical investigator's Brochure. Indianapolis, Ind: Eli Lilly & Company; 1996.
28. Ernst EJ, Klepser ME, Pfaller MA. Postantifungal effects of echinocandin, azole, and polyene antifungal agents against Candida albicans and Cryptococcus neoformans. Antimicrob Agents Chemother 2000;44:1108-11.[PubMed]
29. Ernst EJ, Roling EE, Petzold CR, Keele DJ, Klepser ME. In Vitro Activity of Micafungin (FK-463) against Candida spp.: Microdilution, Time-Kill, and Postantifungal-Effect Studies. Antimicrob Agents Chemother 2002;46:3846-53.[PubMed]
30. Espinel-Ingroff A. Comparison of In vitro activities of the new triazole SCH56592 and the echinocandins MK-0991 (L-743,872) and LY303366 against opportunistic filamentous and dimorphic fungi and yeasts. J Clin Microbiol 1998;36:2950-6. [PubMed]
31. Espinel-Ingroff A. Evaluation of Broth Microdilution Testing Parameters and Agar Diffusion Etest Procedure for Testing Susceptibilities of Aspergillus spp. to Caspofungin Acetate (MK-0991). J Clin Microbiol 2003;41:403-9.[PubMed]
32. Feldmesser M, Kress Y, Mednick A, Casadevall A. The effect of the echinocandin analogue caspofungin on cell wall glucan synthesis by Cryptococcus neoformans. J Infect Dis 2000;182:1791-5.
33. Franzot SP, Casadevall A. Pneumocandin L-743,872 enhances the activities of amphotericin B and fluconazole against Cryptococcus neoformans in vitro. Antimicrob Agents Chemother 1997;41:331-6.[PubMed]
34. Fromtling RA. Micafungin sodium (FK-463). Drugs Today (Barc) 2002;38:245-57.[PubMed]
35. Fromtling RA, Abruzzo GK. L-671,329, a new antifungal agent. III. In vitro activity, toxicity and efficacy in comparison to aculeacin. J Antibiot (Tokyo) 1989;42:174-8.[PubMed]
36. Gallagher JC, Dodds Ashley ES, Drew RH, Perfect JR. Antifungal pharmacotherapy for invasive mould infections. Expert Opin Pharmacother 2003;4:147-64.[PubMed]
37. Garbino J, Lew D, Hirschel B, Rohner P. Caspofungin in the treatment of oropharyngeal candidiasis. Int J Clin Pract 2003;57:143-4. [PubMed]
38. Gonzalez GM, Tijerina R, Najvar LK, Bocanegra R, Luther M, Rinaldi MG, Graybill JR. Correlation between antifungal susceptibilities of Coccidioides immitis in vitro and antifungal treatment with caspofungin in a mouse model. Antimicrob Agents Chemother 2001;45:1854-9. [PubMed]
39. Graybill JR. The echinocandins, first novel class of antifungals in two decades: will they live up to their promise? Int J Clin Pract 2001;55:633-8. [PubMed]
40. Graybill JR, Najvar LK, Bocanegra R, Hector RF, Luther MF. Efficacy of nikkomycin Z in the treatment of murine histoplasmosis. Antimicrob Agents Chemother 1998;42:2371-4.[PubMed]
41. Groll AH, Gullick BM, Petraitiene R, Petraitis V, Candelario M, Piscitelli SC, Walsh TJ. Compartmental pharmacokinetics of the antifungal echinocandin caspofungin (MK-0991) in rabbits. Antimicrob Agents Chemother 2001;45:596-600. [PubMed]
42. Groll AH, Mickiene D, Petraitiene R, Petraitis V, Lyman CA, Bacher JS, Piscitelli SC, Walsh TJ. Pharmacokinetic and pharmacodynamic modeling of anidulafungin (LY303366): reappraisal of its efficacy in neutropenic animal models of opportunistic mycoses using optimal plasma sampling. Antimicrob Agents Chemother 2001;45:2845-55. [PubMed]
43. Hajdu R, Thompson R, Sundelof JG, Pelak BA, Bouffard FA, Dropinski JF, Kropp H. Preliminary animal pharmacokinetics of the parenteral antifungal agent MK-0991 (L-743,872). Antimicrob Agents Chemother 1997;41:2339-44. [PubMed]
44. Ikeda F, Wakai Y, Matsumoto S, Maki K, Watabe E, Tawara S, Goto T, Watanabe Y, Matsumoto F, Kuwahara S. Efficacy of FK463, a new lipopeptide antifungal agent, in mouse models of disseminated candidiasis and aspergillosis. Antimicrob Agents Chemother 2000;44:614-8.[PubMed]
45. Ito M, Nozu R, Kuramochi T, Eguchi N, Suzuki S, Hioki K, Itoh T, Ikeda F. Prophylactic effect of FK463, a novel antifungal lipopeptide, against Pneumocystis carinii infection in mice. Antimicrob Agents Chemother 2000;44:2259-62. [PubMed]
46. James PG, Cherniak R, Jones RG, Stortz CA, Reiss E. Cell-wall glucans of Cryptococcus neoformans Cap 67. Carbohydr Res 1990;198:23-38. [PubMed]
47. Kartsonis N, DiNubile MJ, Bartizal K, Hicks PS, Ryan D, Sable CA. Efficacy of caspofungin in the treatment of esophageal candidiasis resistant to fluconazole. J Acquir Immune Defic Syndr 2002;31:183-7.[PubMed]
48. Keating GM, Jarvis B. Caspofungin. Drugs 2001;61:1121-9; discussion 1130-1. [PubMed]
49. Kohler S, Wheat LJ, Connolly P, Schnizlein-Bick C, Durkin M, Smedema M, Goldberg J, Brizendine E. Comparison of the echinocandin caspofungin with amphotericin B for treatment of histoplasmosis following pulmonary challenge in a murine model. Antimicrob Agents Chemother 2000;44:1850-4.[PubMed]
50. Kontoyiannis DP, Hachem R, Lewis RE, Rivero G, Kantarjian H, Raad II. Efficacy and toxicity of the caspofungin/liposomal amphotericin B combination in documented or possible invasive aspergillosis in patientens with hematologic malignancies. In: 42nd Intersience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego: American Society for Mycrobiology; 2002.
51. Kuhn DM, George T, Chandra J, Mukherjee PK, Ghannoum MA. Antifungal susceptibility of Candida biofilms: unique efficacy of amphotericin B lipid formulations and echinocandins. Antimicrob Agents Chemother 2002;46:1773-80. [PubMed]
52. Kurtz MB, Abruzzo G, Flattery A, Bartizal K, Marrinan JA, Li W, Milligan J, Nollstadt K, Douglas CM. Characterization of echinocandin-resistant mutants of Candida albicans: genetic, biochemical, and virulence studies. Infect Immun 1996;64:3244-51. [PubMed]
53. Kurtz MB, Douglas CM. Lipopeptide inhibitors of fungal glucan synthase. J Med Vet Mycol 1997;35:79-86. [PubMed]
54. Kurtz MB, Heath IB, Marrinan J, Dreikorn S, Onishi J, Douglas C. Morphological effects of lipopeptides against Aspergillus fumigatus correlate with activities against (1,3)-beta-D-glucan synthase. Antimicrob Agents Chemother 1994;38:1480-9.[PubMed]
55. Laverdiere M, Hoban D, Restieri C, Habel F. In vitro activity of three new triazoles and one echinocandin against Candida bloodstream isolates from cancer patients. J Antimicrob Chemother 2002;50:119-23.[PubMed]
56. Luque JC, Clemons KV, Stevens DA. Efficacy of micafungin alone or in combination against systemic murine aspergillosis. Antimicrob Agents Chemother 2003;47:1452-5.[PubMed]
57. Maertens J, Raad I, Petrikkos G, Selleslag D, Petersen F, Sable C, Kartonis N, Ngai A, Taylor A, Patterson TF. Update of the multicenter noncomparative study of caspofungin in adults with invasive aspergillosis refractory or intolerant to other antifungal agents: Analysis of 90 patients. In: 42nd Interscience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego: American Society for Microbiology; 2002.
58. Manavathu EK, Alangaden GJ, Chandrasekar PH. Differential activity of triazoles in two-drug combinations with the echinocandin caspofungin against Aspergillus fumigatus. J Antimicrob Chemother 2003;51:1423-5.[PubMed]
59. Marco F, Pfaller MA, Messer SA, Jones RN. Activity of MK-0991 (L-743,872), a new echinocandin, compared with those of LY303366 and four other antifungal agents tested against blood stream isolates of Candida spp. Diagn Microbiol Infect Dis 1998;32:33-7.[PubMed]
60. Marr KA, Lyons CN, Rustad TR, Bowden RA, White TC, Rustad T. Rapid, transient fluconazole resistance in Candida albicans is associated with increased mRNA levels of CDR. Antimicrob Agents Chemother 1998;42:2584-9.[PubMed]
61. Matsumoto S, Wakai Y, Nakai T, Hatano K, Ushitani T, Ikeda F, Tawara S, Goto T, Matsumoto F, Kuwahara S. Efficacy of FK463, a new lipopeptide antifungal agent, in mouse models of pulmonary aspergillosis. Antimicrob Agents Chemother 2000;44:619-21.[PubMed]
62. Meunier F, Lambert C, Van der Auwera P. In-vitro activity of cilofungin (LY121019) in comparison with amphotericin B. J Antimicrob Chemother 1989;24:325-31.[PubMed]
63. Mikamo H, Sato Y, Tamaya T. In vitro antifungal activity of FK463, a new water-soluble echinocandin-like lipopeptide. J Antimicrob Chemother 2000;46:485-7. [PubMed]
64. Moore CB, D.W. D. Tolerance and fungicidality in vitro of caspofungin, micafungin and anidilafungin against Candida guillermondii. In: 42nd Interscience Conference on Antimicrobial Agents and Chemotherapy; 2002: American Society for Microbiology; 2002.
65. Moore CB, Oakley KL, Denning DW. In vitro activity of a new echinocandin, LY303366, and comparison with fluconazole, flucytosine and amphotericin B against Candida species. Clin Microbiol Infect 2001;7:11-6. [PubMed]
66. Mora-Duarte J, Betts R, Rotstein C, Colombo AL, Thompson-Moya L, Smietana J, Lupinacci R, Sable C, Kartsonis N, Perfect J. Comparison of caspofungin and amphotericin B for invasive candidiasis. N Engl J Med 2002;347:2020-9. [PubMed]
67. Nakai T, Uno J, Ikeda F, Tawara S, Nishimura K, Miyaji M. In Vitro Antifungal Activity of Micafungin (FK463) against Dimorphic Fungi: Comparison of Yeast-Like and Mycelial Forms. Antimicrob Agents Chemother 2003;47:1376-81. [PubMed]
68. National Committee for Clinical Laboratory Standards. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Conidium-Forming Filamentous Fungi (M38-A). Wayne, Pa: National Committee for Clinical Laboratory Standards; 2002.
69. National Committee for Clinical Laboratory Standards. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts: Approved Standard (M27-A). Wayne, Pa: National Committee for Clinical Laboratory Standards; 1997.
70. Onishi J, Meinz M, Thompson J, Curotto J, Dreikorn S, Rosenbach M, Douglas C, Abruzzo G, Flattery A, Kong L, Cabello A, Vicente F, Pelaez F, Diez MT, Martin I, Bills G, Giacobbe R, Dombrowski A, Schwartz R, Morris S, Harris G, Tsipouras A, Wilson K, Kurtz MB. Discovery of novel antifungal (1,3)-beta-D-glucan synthase inhibitors. Antimicrob Agents Chemother 2000;44:368-77.[PubMed]
70a. Osherov N, May GS, Albert ND, Kontoyiannis DP. Overexpression of Sbe2p, a Golgi protein, results in resistance to caspofungin in Saccharomyces cerevisiae. Antimicrob Agents Chemother 2002;46:2462-69. [PubMed]
71. Parent SA, Nielsen JB, Morin N, Chrebet G, Ramadan N, Dahl AM, Hsu MJ, Bostian KA, Foor F. Calcineurin-dependent growth of an FK506- and CsA-hypersensitive mutant of Saccharomyces cerevisiae. J Gen Microbiol 1993;139 ( Pt 12):2973-84.[PubMed]
72. Perea S, Gonzalez G, Fothergill AW, Kirkpatrick WR, Rinaldi MG, Patterson TF. In vitro interaction of caspofungin acetate with voriconazole against clinical isolates of Aspergillus spp. Antimicrob Agents Chemother 2002;46:3039-41. [PubMed]
73. Petraitis V, Petraitiene R, Sarafandi AA, Kelaher AM, Lyman CA, Casler HE, Sein T, Groll AH, Bacher J, Avila NA, Walsh TJ. Combination Therapy in Treatment of Experimental Pulmonary Aspergillosis: Synergistic Interaction between an Antifungal Triazole and an Echinocandin. J Infect Dis 2003;187:1834-43. [PubMed]
74. Pfaller M, Riley J, Koerner T. Effects of cilofungin (LY121019) on carbohydrate and sterol composition of Candida albicans. Eur J Clin Microbiol Infect Dis 1989;8:1067-70.[PubMed]
75. Pfaller MA, Diekema DJ, Messer SA, Hollis RJ, Jones RN. In Vitro Activities of Caspofungin Compared with Those of Fluconazole and Itraconazole against 3,959 Clinical Isolates of Candida spp., Including 157 Fluconazole-Resistant Isolates. Antimicrob Agents Chemother 2003;47:1068-71. [PubMed]
76. Pfaller MA, Jones RN, Doern GV, Fluit AC, Verhoef J, Sader HS, Messer SA, Houston A, Coffman S, Hollis RJ. International surveillance of blood stream infections due to Candida species in the European SENTRY Program: species distribution and antifungal susceptibility including the investigational triazole and echinocandin agents. SENTRY Participant Group (Europe). Diagn Microbiol Infect Dis 1999;35:19-25. [PubMed]
77. Pfaller MA, Marco F, Messer SA, Jones RN. In vitro activity of two echinocandin derivatives, LY303366 and MK-0991 (L-743,792), against clinical isolates of Aspergillus, Fusarium, Rhizopus, and other filamentous fungi. Diagn Microbiol Infect Dis 1998;30:251-5. [PubMed]
78. Powles MA, Liberator P, Anderson J, Karkhanis Y, Dropinski JF, Bouffard FA, Balkovec JM, Fujioka H, Aikawa M, McFadden D, Schmatz D. Efficacy of MK-991 (L-743,872), a semisynthetic pneumocandin, in murine models of Pneumocystis carinii. Antimicrob Agents Chemother 1998;42:1985-9. [PubMed]
79. Powles R, Chopra R. Assessment of maximum tolerated dose (MTD) And pharmacokinetics (PK) of FK463 in neutropenic patients after haematopoetic stem cell transplantation (abstract 5217). Blood 2001;98:361b.
80. Qadota H, Python CP, Inoue SB, Arisawa M, Anraku Y, Zheng Y, Watanabe T, Levin DE, Ohya Y. Identification of yeast Rho1p GTPase as a regulatory subunit of 1,3-beta-glucan synthase. Science 1996;272:279-81. [PubMed]
81. Ramage G, VandeWalle K, Bachmann SP, Wickes BL, Lopez-Ribot JL. In vitro pharmacodynamic properties of three antifungal agents against preformed Candida albicans biofilms determined by time-kill studies. Antimicrob Agents Chemother 2002;46:3634-6.[PubMed]
82. Ramesh MS BI, Ganesan LT. Postantifungal effect of older and newer antifungal agents on Aspergillus fumigatus and Candida albicans. In: 41st Interscience Conference on Antimicrobial Agents and Chemotherapy; 2001; Chicago: American Society for Microbiology; 2001.
83. Roberts J, Schock K, Marino S, Andriole VT. Efficacies of two new antifungal agents, the triazole ravuconazole and the echinocandin LY-303366, in an experimental model of invasive aspergillosis. Antimicrob Agents Chemother 2000;44:3381-8. [PubMed]
84. Rubin MA, Carroll KC, Cahill BC. Caspofungin in combination with itraconazole for the treatment of invasive aspergillosis in humans. Clin Infect Dis 2002;34:1160-1.[PubMed]
85. Sanglard D, Ischer F, Monod M, Bille J. Cloning of Candida albicans genes conferring resistance to azole antifungal agents: characterization of CDR2, a new multidrug ABC transporter gene. Microbiology 1997;143 ( Pt 2):405-16.[PubMed]
86. Sanglard D, Kuchler K, Ischer F, Pagani JL, Monod M, Bille J. Mechanisms of resistance to azole antifungal agents in Candida albicans isolates from AIDS patients involve specific multidrug transporters. Antimicrob Agents Chemother 1995;39:2378-86. [PubMed]
87. Sanglard D, Odds FC. Resistance of Candida species to antifungal agents: molecular mechanisms and clinical consequences. Lancet Infect Dis 2002;2:73-85. [PubMed]
88. Schuetzer-Muehlbauer M, Willinger B, Krapf G, Enzinger S, Presterl E, Kuchler K. The Candida albicans Cdr2p ATP-binding cassette (ABC) transporter confers resistance to caspofungin. Mol Microbiol 2003;48:225-35.[PubMed]
89. Seibel N, Schwartz C, Arrieta A, Flynn P, Shad A, Albano E, Walsh TJ. A phase I study to determine the safety and pharmacokinetics (PK) of FK463 (echinocandin) in febrile neutropenic pediatric patients. In: 40nd Intersience Conference on Antimicrobial Agents and Chemotherapy; 2000; Toronto, Ontario, Canada: American Society for Mycrobiology; 2000.
90. Serrano Mdel C, Valverde-Conde A, Chavez M M, Bernal S, Claro RM, Peman J, Ramirez M, Martin-Mazuelos E. In vitro activity of voriconazole, itraconazole, caspofungin, anidulafungin (VER002, LY303366) and amphotericin B against aspergillus spp. Diagn Microbiol Infect Dis 2003;45:131-5.[PubMed]
91. Shalit I, Shadkchan Y, Samra Z, Osherov N. In Vitro Synergy of Caspofungin and Itraconazole against Aspergillus spp.: MIC versus Minimal Effective Concentration End Points. Antimicrob Agents Chemother 2003;47:1416-8.[PubMed]
92. Shematek EM, Braatz JA, Cabib E. Biosynthesis of the yeast cell wall. I. Preparation and properties of beta-(1 leads to 3)glucan synthetase. J Biol Chem 1980;255:888-94. [PubMed]
93. Slaven JW, Anderson MJ, Sanglard D, Dixon GK, Bille J, Roberts IS, Denning DW. Increased expression of a novel Aspergillus fumigatus ABC transporter gene, atrF, in the presence of itraconazole in an itraconazole resistant clinical isolate. Fungal Genet Biol 2002;36:199-206.[PubMed]
94. Sora F, Chiusolo P, Piccirillo N, Pagano L, Laurenti L, Farina G, Sica S, Leone G. Successful treatment with caspofungin of hepatosplenic candidiasis resistant to liposomal amphotericin B. Clin Infect Dis 2002;35:1135-6.[PubMed]
95. Stone EA, Fung HB, Kirschenbaum HL. Caspofungin: an echinocandin antifungal agent. Clin Ther 2002;24:351-77; discussion 329.[PubMed]
96. Stone JA, Holland SD, Wickersham PJ, Sterrett A, Schwartz M, Bonfiglio C, Hesney M, Winchell GA, Deutsch PJ, Greenberg H, Hunt TL, Waldman SA. Single- and multiple-dose pharmacokinetics of caspofungin in healthy men. Antimicrob Agents Chemother 2002;46:739-45. [PubMed]
97. Tawara S, Ikeda F, Maki K, Morishita Y, Otomo K, Teratani N, Goto T, Tomishima M, Ohki H, Yamada A, Kawabata K, Takasugi H, Sakane K, Tanaka H, Matsumoto F, Kuwahara S. In vitro activities of a new lipopeptide antifungal agent, FK463, against a variety of clinically important fungi. Antimicrob Agents Chemother 2000;44:57-62. [PubMed]
98. Thompson JR, Douglas CM, Li W, Jue CK, Pramanik B, Yuan X, Rude TH, Toffaletti DL, Perfect JR, Kurtz M. A glucan synthase FKS1 homolog in cryptococcus neoformans is single copy and encodes an essential function. J Bacteriol 1999;181:444-53.[PubMed]
99. Thye D, Kilfoil G, Kilfoil T, Henkel T. Anidulafungin: Pharmacokinetics in subjects with severe hepatic impairment. In: 42nd Intersience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego: American Society for Mycrobiology; 2002.
100. Thye D, Kilfoil T, Kilfoil G, Henkel T. Anidulafungin: Safety and pharmacokinetics in subjects reiceiving concomitant cyclosporine. In: 42nd Intersience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego: American Society for Mycrobiology; 2002.
101. Thye D, Marbury T, Kilfoil G, Kilfoil T, Henkel T. Anidulafungin: Pharmacokinetics in subjects receiving hemodialysis. In: 42nd Intersience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego: American Society for Mycrobiology; 2002.
102. Thye D, Marbury T, Kilfoil G, Kilfoil T, Henkel T. Anidulafungin: Pharmacokinetics in subjects with renal impairment. In: 42nd Intersience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego: American Society for Mycrobiology; 2002.
103. Townsend R, Herbert M, Dessimoz M, Wisemandle W, Bekersky I. Pharmacokinetics (PK) of micafungin, an echinocandin antifungal, in subjects with severe renal dysfunction. In: 31st American College of Clinical Pharmacology; 2002; San Francisco: American College of Clinical Pharmacology; 2002.
104. Townsend R, Herbert M, Dessimoz M, Wisemandle W, Bekersky I. Pharmacokinetics of micafungin, an echinocandin antifungal, in subjects with moderate hepatic dysfunction. In: 31st American College of Clinical Pharmacology; 2002; San Francisco: American College of Clinical Pharmacology; 2002.
105. Townsend R, Herbert M, Wisemandle W, Bekersky I. Concomitant pharmacokinetics (PK) of micafungin, and echinocandin antifungal, and cyclosporine in healthy volunteers. In: 31st American College of Clinical Pharmacology; 2002; 2002; San Francisco: American College of Clinical Pharmacology; 2002.
106. van Burik J, Ratanatharathorn V, Lipton J, Miller C. Randomized, double-blind trial of micafungin versus fluconazole for prophylaxis of invasive fungal infections in patients undergoing hemopoietic stem cell transplant, NIAID/BAMSG Protocol 46. In: 42nd Intersience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego: American Society for Mycrobiology; 2002.
107. van Duin D, Casadevall A, Nosanchuk JD. Melanization of Cryptococcus neoformans and Histoplasma capsulatum reduces their susceptibilities to amphotericin B and caspofungin. Antimicrob Agents Chemother 2002;46:3394-400. [PubMed]
108. Vazquez JA, Lynch M, Boikov D, Sobel JD. In vitro activity of a new pneumocandin antifungal, L-743,872, against azole-susceptible and -resistant Candida species. Antimicrob Agents Chemother 1997;41:1612-4. [PubMed]
109. Vazquez JA, Nagappan V, Boikov D. In vitro evaluation of caspofungin acetate (Cancidas) against Candida glabrata: MICs, MFCs, time kill and synergy assays. In: Interscience Conference on Antimicrobial Agents and Chemotherapy; 2002; San Diego; 2002.
110. Verweij PE, Oakley KL, Morrissey J, Morrissey G, Denning DW. Efficacy of LY303366 against amphotericin B-susceptible and -resistant Aspergillus fumigatus in a murine model of invasive aspergillosis. Antimicrob Agents Chemother 1998;42:873-8. [PubMed]
111. Villanueva A, Gotuzzo E, Arathoon EG, Noriega LM, Kartsonis NA, Lupinacci RJ, Smietana JM, DiNubile MJ, Sable CA. A randomized double-blind study of caspofungin versus fluconazole for the treatment of esophageal candidiasis. Am J Med 2002;113:294-9. [PubMed]
112. Warn PA, Sharp A, Morrissey G, Denning DW. In vivo activity of micafungin in a persistently neutropenic murine model of disseminated infection caused by Candida tropicalis. J Antimicrob Chemother 2002;50:1071-4.[PubMed]
113. Watabe E, Nakai T, Matsumoto S, Ikeda F, Hatano K. Killing Activity of Micafungin against Aspergillus fumigatus Hyphae Assessed by Specific Fluorescent Staining for Cell Viability. Antimicrob Agents Chemother 2003;47:1995-8. [PubMed]
Tables
Table 1. In vitro activity of caspofungin, micafungin and anidulafungin against Candida sp. (9, 22, 28, 29, 59, 63, 65, 75, 76, 97)
| Caspofungin | Micafungin | Anidulafungin | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Species | N | MIC (Range) | MIC90 | n | MIC (Range) | MIC90 | n | MIC (Range) | MIC90 |
| Candida albicans | 3450 | 0.007->0.8 | 0.125-0.5 | 45 | <0.0039-0.0625 | 0.0156-0.0625 | 42 | <0.001-0.015 | 0.0078 |
| Candida glabrata | 756 | 0.007->0.8 | 0.25-1 | 39 | 0.0078-0.0625 | 0.0156-0.0039 | 13 | 0.0039-0.03 | 0.03 |
| Candida krusei | 185 | 0.125-2 | 1 | 19 | 0.125-0.25 | 0.125-0.25 | 11 | 0.0078-0.015 | 0.015 |
| Candida parapsilosis | 635 | 0.03->16 | 1-4 | 29 | 0.5-2 | 1 | 12 | 0.125-16 | 1 |
| Candida tropicalis | 127 | 0.03-2 | 0.125-1 | 13 | 0.0156-0.125 | 0.0313-0.125 | 10 | 0.002-0.015 | 0.0015 |
| Candida lusitaniae | 35 | 0.12-2 | 1-2 | 3 | 0.0015 | 0.0015 | |||
MIC = minimum inhibitory concentration (µg/ml); MIC90, = MIC required to inhibit 90% of organisms compared to growth control.
MICs were determined by broth microdilution with RPMI-1640 medium after incubation for 48 hours.
Table 2. In vitro activity of caspofungin, micafungin and anidulafungin against Aspergillus sp. (8, 30, 31, 70, 83, 90, 97)
| Caspofungin | Micafungin | Anidulafungin | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Species | n | MIC90 | n | MEC (Range) | n | MIC (Range) | MIC90 | n | MIC90 |
| Aspergillus flavus | 97 | 0.12->32 | 58 | <0.03-2 | 14 | 0.0078-0.0156 | 0.0156 | 9 | <0.03 |
| Aspergillus fumigatus | 274 | 0.25->32 | 191 | <0.03->8 | 47 | 0.0078-0.0156 | 0.0078-0.0156 | 28 | <0.03 |
| Aspergillus niger | 30 | 0.25-1 | 39 | <0.03-1 | 16 | <0.0039-0.0156 | 0.0078 | 9 | <0.03 |
| Aspergillus terreus | 36 | 0.2-0.6 | 26 | <0.03-0.5 | 8 | <0.0039-0.0078 | 0.0078 | 2 | <0.03 |
MIC = minimum inhibitory concentration (µg/ml); MIC90, = MIC required to inhibit 90% of organisms compared to growth controls.
MEC = minimal effective concentration
MICs were determined by broth microdilution with RPMI-1640 medium after incubation for 48 hours.
Table 3. Pharmacokinetic parameters of caspofungin, micafungin and anidulafungin (16, 27, 34, 48, 79, 95, 96, 102, 103, 105)
| Pharmacokinetic Parameter | Caspofungin | Micafungin | Anidulafungin |
|---|---|---|---|
| Plasma elimination half-life | 9-11 h | 11.6-15.2 h | 29 h 30 h* |
| Volume of distribution | 9.67L | 14L | 41.2L |
| Plasma protein binding | Extensive (97%) | Extensive (99%) | 84% |
| Metabolism | Hepatic (primarily hydrolysis to inactive metabolites) | Hepatic | Hepatic |
| Clearance | 12 mL/min | 13 mL/min | 16 mL/min |
* After oral dosing
Figure 1. Structure of (a) Caspofungin, (b) Micafungin, and (c) Anidulafungin.
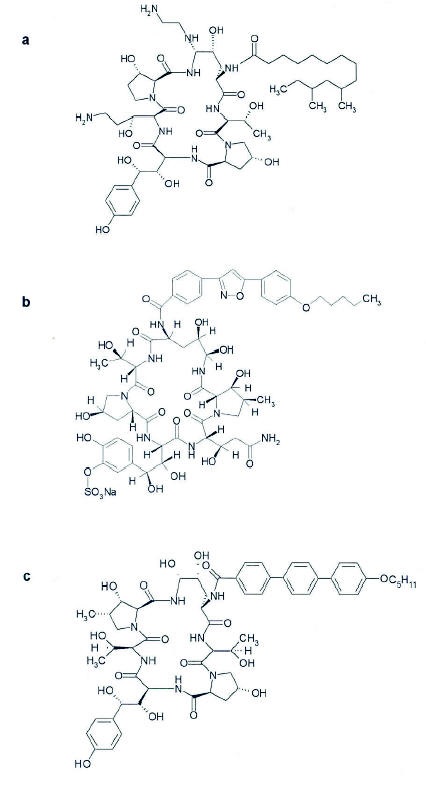
Figure 2. Effects of in vitro echinocandin exposure to (a) Candida species, (b) Aspergillus species.
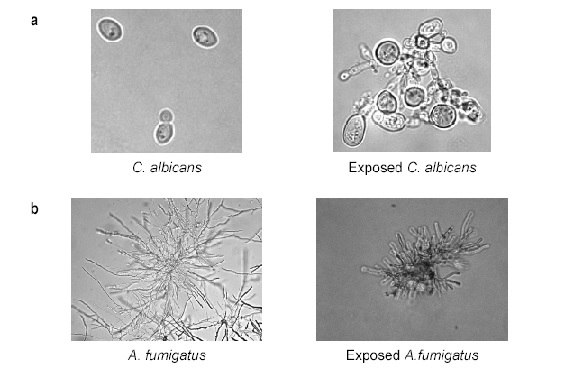
Note: While the drugs typically kill Candida species, potentially resulting in fungicidal effects, activity against Aspergillus species is only appreciated in growing cells located at branch points and tips of filaments. latter effect causes ballooning, blunting, and other forms of aberrant growth upon microscopic evaluation (b).
