Treponema Pallidum (Syphilis)
Authors: Lori E. Fantry, MD, MPH, Edmund C. Tramont, M.D., M.A.C.P
GENERAL INFORMATION
Syphilis has been known to afflict mankind at least since the discovery of the "New World" by Christopher Columbus in 1492 . Before the advent of antibiotics, it was one of the most common infections, afflicting up to 10% of the adult populations in the Western World. Although it was one of the first infections to be treated successfully with antibiotics (118), debate remains with regards to what constitutes optimal treatment. The fundamental reason for this controversy is the inability to culture Treponema pallidum in vitro on routine culture media or in tissue culture. The only means of laboratory culture remains in laboratory animals. This requirement has made it extremely difficult to correlate clinical signs and symptoms with the presence or absence of replicating spirochetes or perform simple in vitro antimicrobial susceptibility testing which in turn has forced clinicians to rely on imperfect serological tests to diagnose syphilis and gage the effectiveness of therapy.
MICROBIOLOGY
Syphilis is caused by a thin, tightly coiled spirochete, Treponema pallidum subspecies pallidum (119). It is microaerophilic and cannot grow on standard culture media. It is a member of the family Spirochaeticea and is related to other spirochete genera which have the capacity to infect man, namely Borrelia and Leptospira. Other pathogenic treponemes for man include T. pallidum subspecies pertenue, the causative agent of yaws; T. carateum, the causative agent of pinta; and T. pallidum subspecies endemicum, the agent associated with non-venereal or endemic syphilis.
The entire genome of T. pallidum has been completely sequenced (36). It is a small genome of only 1,138,006 base pairs and 1041 predicted coding sequences (open reading frames). Because of this, it lacks many pathways including the tricarboxylic acid cycle, components of oxidative phosphorylation, and most biosynthetic pathways and relies on the host to perform necessary functions (89).
EPIDEMIOLOGY
Although syphilis can be spread by passage through the birth placenta (congenital syphilis), by kissing or other close contact with an active lesion, transfusion of fresh human blood, or by direct inoculation, the vast majority of cases are transmitted by sexual contact (119). Persons are most infectious early in disease when a chancre, mucous patch, or condyloma latum is present and by 4 years after acquiring the disease, an untreated immunocompetent person is essentially non-infectious. Reinfection is possible and, in fact, not uncommon (34).
The disease is worldwide in distribution and the World Health Organization's (WHO) estimates that there were 10.6 million new cases of syphilis in 2008 (125). It occurs primarily in persons between the ages of 15 and 40 years but about 1 million infants are born each year with congenital syphilis.
In the U.S., prior to the advent of penicillin treatment, up to 10% of persons living in urban areas were infected (118). The incidence plummeted to very low levels by the mid-1950s and kept decreasing until the year 2000 when the incidence reached an all- time low (13). In fact, in October of 1999, U.S. Surgeon General David Satcher announced the Center for Disease Control and Prevention's (CDC) National Plan to Eliminate Syphilis (10).
However, since 2000 there has been an annual increase in the rate of primary and secondary syphilis in the U.S. (11). This increase is primarily fueled by increases among men who have sex with men although cases in women also increased from 2004-2008 (15). The sero-prevalence is greatest in the South Eastern U.S., urban areas, among African Americans, and in persons with lower income and less education (42).
CLINICAL MANIFESTATIONS
The clinical disease manifestations have been well characterized for over 100 years and are traditionally divided into five stages: incubating, primary, secondary, latent (early latent and late latent), and late or tertiary syphilis (neurosyphilis, cardiovascular syphilis and gummatous syphilis) (119).
Primary syphilis is the stage of infection which occurs 3 to 90 days (a median of 3 weeks) after infection. It is most commonly characterized by a single, painless chancre or ulcer that develops at the site of inoculation ( Figure 1) (119). The chancre typically has a smooth base with raised and firm borders. However, in some persons, it does not develop at all; in other persons, it is so small that it may go completely unnoticed; and in others, especially those with human immunodeficiency virus (HIV) infection, multiple ulcers may develop (101, 102). The untreated lesion or lesions usually heal spontaneously in 2 to 8 weeks (range 1 to 12 weeks).
Secondary syphilis, the result of the interaction between a large spirochete load and the host's immune response, is the stage of infection in which there is widespread dissemination to various parts of the body. It becomes evident a mean of 6 weeks (range 2 to 16 weeks) after inoculation. In 90% of cases, there is a rash which most commonly is widely disseminated, maculopapular and involves the palms and soles but other dermatological manifestations are also common (Figure 2). In addition, over 50% of cases have fever, malaise, anorexia, weight loss, pharyngitis, laryngitis, and/or arthralgias. Other clinical manifestations include lesions in the mouth and oral cavity, lymphadenopathy, condyloma latum, glomerulonephritis, nephritic syndrome, hepatitis, arthritis, osteitis, and periosteitis.
Latent syphilis is the period of months to years post infection in which there are no outward clinical manifestations of disease despite viable organisms. Clinical relapses can occur during the first year of the latent stage (referred to as the early latent phase) and is felt to be the result of waning specific cellular immunity.
Tertiary or late syphilis occurs in up to 35% of untreated patients ten to twenty-five years after the initial infection. Late syphilis can be categorized into neurosyphilis, cardiovascular syphilis, and granulomatous syphilis. The latter two forms are uncommon in the antibiotic era due to the frequent exposure to antibiotics while today t neurosyphilis is the most common manifestation of tertiary/late syphilis because of the generally poor penetration of antibiotics into the CNS.
LABORATORY DIAGNOSIS
Because of our inability to culture T. pallidum using standard laboratory methods, a variety of tests have been developed to try to overcome this shortcoming (Table 1).
DIRECT DETECTION METHODS
Direct detection methods are not widely available and require specialized laboratories with trained technicians. Hence, false negative tests are relatively common, about 30%.
Dark Field Microscopy
Dark field microscopy remains a sensitive, direct, and quick method for diagnosing syphilis in active lesions (119). However, it requires a special dark field equipped microscope and skilled technicians that are not usually available at most medical facilities today, including many large hospitals in the U.S. Specimens for microscopic examination are best obtained from serous transudate of moist lesions such as such as a primary chancre, condyloma latum (wart like lesions, sometimes extensive), or mucous patches (shallow ulcers on mucous membranes, non-painful unless secondarily infected). However, they also can be collected from dry skin or lymph nodes by non-bactericidal saline aspiration. When obtaining a sample, the surface should be cleaned with non-bactericidal saline and gently abraded with dry gauze, just enough so that a few scattered red blood cells are seen on the slide. The cleaning should be performed without soap, a topical antiseptic or bactericidal saline because dead and non-motile organisms are difficult to visualize. The specimen should be placed on a glass slide with a cover slip placed on top. When using serous exudates, a drop of non-bactericidal saline may be added if the serous preparation is too thick. Under dark field microscopy, T. pallidum will appear as corkscrew shape in spiraling motion with a 90° undulation about its midpoint. At least three specimens should be examined before deciding that a lesion is non-syphilitic.
Polymerase chain reaction (PCR)
PCR can also be used to detect T. pallidum genetic material, most often the PolA gene (38, 44, 49, 51, 60, 64, 68, 92). To date, it is most commonly used to diagnose congenital syphilis. However, it has also been shown to be effective at diagnosing primary syphilis with sensitivities between 73% and 95% and specificities 95% (44, 51, 92). For the diagnosis of congenital syphilis, frozen tissue and formalin-fixed, paraffin-embedded tissue and, for the diagnosis of primary syphilis, swabs of the ulcer can be submitted to the CDC for processing with prior approval from the local health department (9). Other specimen types are acceptable but have less diagnostic value. Sensitivities of blood samples in primary syphilis are as low as 18% (44). The web site for submission to the CDC is http://www.cdc.gov/laboratory/specimen-submission/detail.html?CDCTestCode=CDC-10176.
Immunofluorescent and Immunoperoxidase Antibody Staining
Specific immunofluorescent or immunoperoxidase antibody staining can be used to visualize organisms, including nonviable spirochetes, from mucocutaneous lesions, lymph nodes, or dry skin (19, 119). In addition, it can be used for examining non-frozen biopsy material (118).
SEROLOGICAL TESTS
Nontreponemal Reaginic Antibody Tests
Syphilis reaginic antibodies are IgG and IgM antibodies directed against a lipoidal antigen resulting from the interaction of host tissues with T. pallidum or from T. pallidum itself (119). The earliest cardiolipin antigens used to measure reaginic antibody were crude extracts from beef livers or hearts and false-positive tests were common. However, today's preparation, the cardiolipin-cholesterol-lecithin, is much purer and hence there are less false-positive reactions. The nontreponemal test first developed was the Venereal Disease Research Laboratory (VDRL) slide test (118). In this test, serum is heated to 56° C and tested to see if it can flocculate a suspension of a cardiolipin-cholesterol-lecithin antigen. Except for its use in diagnosing neurosyphilis, it has now been largely replaced by the modifications such as the rapid plasma reagin (RPR) card test, automated reagin test (ART), and the toluidine red unh<peated test (TRUST). In addition, there is also now a modified RPR test that can be done without requiring a laboratory at the point of care (121).
Nontreponemal tests become positive shortly after initial infection, peak during the secondary or early latent stage, and then decline with time (48) (Table 2). In primary syphilis, an antibody response may not yet have been generated so nontreponemal tests may be negative. In secondary syphilis, virtually 100% of infected persons have positive serological tests for syphilis but in some patients the titers are so high that the test is reported as negative due to the prozone phenomenon (119). If the clinical index of suspicion is high, appropriate serum dilutions should be performed. In latent and late syphilis, titers decline, usually to < 1:4, and actually become negative in 25% of untreated persons.
On the other hand, whenever there is a strong immunologic stimulus (e.g., acute bacterial or viral infection, vaccination, HIV infection), a "false" positive occurs (62, 119). In addition, persons who use injection drugs, have autoimmune or connective tissue diseases (especially systemic lupus erythematosus), or hypergammaglobulinemic states may have "false" positive results. These persons often also have blood tests positive for other factors frequently associated with autoimmune disease such as antinuclear, antithyroid, or antimitochondrial antibodies; rheumatoid factor; and cryoglobulins. A negative specific treponemal test will confirm that the test is a false positive and that syphilis can be excluded.
Fluorescent Treponemal Antibody-Absorption (FTA-abs)
The FTA-abs uses. T. pallidum harvested from rabbit testes as the antigen in a standard indirect immunofluorescent antibody test. The first step is to remove so called "natural" cross-reacting antibody that may have been raised against saprophytic treponemes of the oral cavity or genital tract by absorbing the patient's serum with nonpathogenic treponemal antigen (referred to as "sorbent"). The next step is to place the patient's "absorbed" serum on a slide which contains that has fixed T. pallidum as the antigen. If specific antibody to T. pallidum is in the patient's serum, then it is detected when fluorescein-labeled antihuman gamma globulin is added to the slide and examined under a fluorescence microscope. Its interpretation can be quite subjective. This test is used to confirm or refute a positive nontreponemal test. If there is a high index of suspicion, it also is used to make a diagnosis of syphilis even when a nontreponemal test is negative.
T. pallidum Haemagglutination Assay (TPHA) and Microhemagglutination Assay for Antibodies to T. pallidum (MHATP)
The TPHA also measures specific treponemal antibody. It is easier to perform than the FTA-abs and is as specific but not as sensitive, especially in early disease. The MHATP test is similar to the TPHA test except it uses a microtiter plate. "Sorbent" is always used to increase its specificity.
Enzyme-linked Immunosorbent Assay (ELISA)
The syphilis ELISAs are automated tests that use a technique called a qualitative sandwich immunoassay to detect T. pallidum specific antibodies. An ELISA may detect only IgG or IgM but most assays are polyvalent. As with all ELISAs, an enzyme is conjugated with anti-human antibodies and only those wells that contain T. pallidum specific antibodies conjugated to the enzyme will exhibit a color change. The sensitivity and specificity of ELISAs are similar to TPHAs and FTA-Abs (8). When compared with the sensitivities of the RPR test and the MHA-TP, ELISA is more sensitive in all stages of syphilis except in secondary syphilis when all tests show 100% sensitivity (8). The TP ELISA tests are the most common test for syphilis antibodies in the USA.
Immunochromatographic Membrane Test (ICT)
This is a rapid test that detects T. pallidum antibodies employing anti-human immunoglobulins gold conjugate and highly purified TP recombinant proteins bound to a membrane. The sensitivity and specificity are both greater than 98% (20). It has an advantageous over an ELISA in that it is visually read and can be performed at the point of care. With minimum training, health care professionals and other staff can give patients test results in 10-15 minutes at a physician's office, emergency room, or clinic. In 2104, the U.S. Food and Drug Administration (FDA) granted a Clinical Laboratory Improvement Amendments (CLIA) waiver so that one such test, the Syphilis Health Check, can be done at the point of care (120). Other point-of-care tests will likely be approved.
IImmunochromatographic Strip (ICS)
The ICS test is "lateral flow" test in which antibodies in a specimen are detected by becoming bound to antigens, marked with dye, on a cellulose strip. Sensitivity and specificity are both 85% to 98% (50). Like ICTs, it it requires no special training to read, laboratory equipment to run, or refrigeration of reagents or samples. Results can be given in less than 30 minutes at the point of care. Currently, it is only used outside the U.S., primarily i in developing countries, since there is no CLIA waiver.
Line immunoassay (LIA)
The LIA uses recombinant and synthetic polypeptide antigens derived from T. pallidum proteins to determine if a clinical specimen has treponemal antibodies (57). In a recent study, the sensitivity and specificity of LIA were 100 % and 99 %, respectively. Like ICTs and the ICS test, it is inexpensive, rapid, and requires no special laboratory equipment or highly trained personnel so it is well suited for use in developing countries.
Testing Algorithms for Syphilis in Adults except Neurosyphilis
The tradition algorithm for diagnosing syphilis is to screen with a nontreponemal test and then if the nontreponemal test is reactive, verify with a specific tests such as the FTA-abs, TPHA, MHATP, or ELISA. If both tests are reactive, then a person is considered to have present or past syphilis infection. Treatment decisions are based on past history and nontreponemal titers.
Many health care facilities in the U.S. now use treponemal ELISA and will use for screening, so-called reverse sequence screening, because these tests can be automated, thus reducing the need for highly trained technicians.. Since long lived antibodies are detected , confirmation of current active disease requires a nontreponemal test (14).
As with the traditional method, when a person is reactive to both a specific treponemal such as a positive ELISA t and nontreponemal test such as a RPR, then a person is considered to have active disease requiring treatment. When a person is reactive to the treponemal test but nonreactive to the non-treponemal test such as a RPR test, persons with a history of previous treatment will require no further management. Those with no prior history of treatment should have a different treponemal test performed such as an FTA-abs. If the second test is also nonreactive, then the clinician needs to use his/her clinical judgment to decide whether or not a third treponemal test is indicated or to treat empirically.
Testing Algorithms for Congenital Syphilis
The diagnosis of congenital syphilis is best made by testing the mother at the time of birth since infant serum titers, even when the infant is infected, may be non-reactive, especially if the mother has low titers or the mother was infected late in pregnancy (12, 119). If the mother has reactive syphilis serology, then the infant's serum should be evaluated with a RPR or VDRL. Infants should also have a physical exam and dark field microscopy or direct fluorescent staining of any suspicious lesions and radiological and ultrasound studies (12). The placenta or umbilical cord should also be examined using specific fluorescent anti-treponemal antibody staining. IgM-specific antibodies [ELISA, reverse enzyme-linked immunospot (Relispot), FTA-abs, or immunoblotting/Western blot] and PCR are also recommended to make the diagnosis (16, 76, 78, 110, 119). Any positive test requires treatment except if the infant's nontreponemal serology is the same or less than fourfold that of the mother, the mother was known to have been adequately treated with penicillin during pregnancy, and all other tests are negative.
Testing Algorithms for Neurosyphilis
A lumbar puncture with the CSF sent for VDRL, cytology, and protein is the most commonly used method for making the diagnosis (12). In the appropriate clinical setting, a reactive CSF-VDRL is considered diagnostic of syphilis while a negative test does not rule out disease, e.g., it is a specific but not very sensitive test (119). If the CSF-VDRL is negative, a finding of more than five mononuclear cells per cubic millimeter, a protein value of 46 mg/dL or greater, or a glucose of 45 mg/dL or less all are suggestive of neurosyphilis (24, 71). However, the limitations of these tests have been well known for over 90 years and were most recently dramatically illustrated in a 1988 study (71). Four of twelve patients (33%) in whom T. pallidum was cultured from the CSF using rabbits had normal cellular and protein studies as well as a non-reactive CSF-VDRL (RPR) attesting to the low sensitivity for all of these tests, even in combination. In addition, there were a significant number of patients in whom T. pallidum was not isolated from the CSF despite CSF abnormalities including four patients with reactive VDRL's. A serum VDRL (RPR) titer greater than 1:32 most closely correlates with a positive T. pallidum isolation in rabbits (culture) and this was found most commonly in secondary syphilis. Other methods with greater sensitivity and specificity than the CSF-VDRL are the intrathecal T. pallidum antibody (ITPA) and TPHA indices but these are seldom used in clinical practice (119). A FTA-abs test is usually not performed on CSF because a positive test may represent passive transfer of antibody from serum to the CSF and not active CNS disease (119). However, it is highly sensitive and thus a negative test can be helpful in ruling out neurosyphilis, especially in HIV infected patients who often have white blood cells in the CSF. Marra and co-workers found it, along with a determination of a percent CSF cells that were B lymphocytes, to be useful in excluding or establishing a diagnosis of neurosyphilis (75). Clinical judgment must be the deciding factor determining whether or not to treat for neurosyphilis.
PATHOGENESIS
Infection begins when T. pallidum penetrates the host, usually through intact or abraded mucous membranes (119). Although the exact infectious dose in humans is not known, in rabbits, an infectious inoculum can be as few as four spirochetes. The incubation period is directly proportional to the size of the inoculum since clinical lesions do not appear until a concentration of approximately 107 organisms per mg of tissue in rabbits is reached (27). Potential virulence factors which account for T. pallidum's ability to cause disease include several hemolysins, a membrane protein that allows for permeability of nutrients but inaccessible to antibody, and ligands that allow cytoadhesion (36, 49, 93). As with most other infections, it is the ensuing inflammation that is responsible for most of the disease pathology (119).
In early disease, spirochetes can be found in the chancre, the usual first manifestation of syphilis. Invasion into the bloodstream and lymphatics occurs within hours to days of penetration of T. pallidum as evidenced by the fact that patients who received blood transfusions from syphilitic donors in the seronegative incubation period have become infected (119). All organs of the body can be invaded but the skin, lymph nodes, and the central nervous system (CNS) are the sites most often invaded. In the skin, T. pallidum is found in the dermal-epidermal junction zone or throughout the dermis (28). Up to 40% of patients in primary and secondary syphilis have evidence of CNS invasion, including the eye, as evidenced by either abnormal laboratory tests or direct culture of the treponemes in laboratory animals. Late stage disease is most often manifested by invasion of the vasa vasorum of the aorta and/or the arteries of the CNS by T. pallidum (119). In addition, gummas, granulomatous lesions with coagulated or amorphous centers, form most commonly in the skin, liver, bones, and spleen.
Both humoral and cell mediated immune responses are mounted against T. pallidum. At all stages of infection, there are local cellular infiltrates consisting of lymphocytes, macrophages, and plasma cells at the sites of disease (93). In primary syphilis, CD4+ T cells and macrophages are the predominate cell type while in secondary syphilis CD8+ cells predominate. In both primary and secondary syphilis, there is increased expression of Th1 cytokines IL-2 and IFN-gamma. Cell mediated and humoral immune responses peak in secondary syphilis.
Spirochetes may remain alive and continue to replicate in immunologically sequestered sites in the body even with a brisk initial immune response and no outward clinical manifestations of disease in at least 1 of 4 persons who are not treated. In late latent syphilis, after many years of latency, treponemes begin to multiple and Th1 lymphocytes produce high levels of nitric oxide and IL-12 instead of IL-2 and IFN (72). This immunological escape by T. pallidum is thought to be due to three main characteristics of T. pallidum. First of all, it has a very slow dividing time of 30-33 hours. Secondly, it has a scarcity of protruding antigenic proteins membrane proteins which make an antibody response more difficult. Thirdly, it has the ability to vary its antigens by gene conversion which may result in a subpopulation of T. pallidum to immunological defenses such as macrophage phagocytosis (93, 119).
SUSCEPTIBILITY IN VITRO AND IN VIVO
Single Drug
Early susceptibility testing of various antibiotics against T. pallidum in experimental rabbit models established the principles of anti-treponemal syphilis therapy. Using the rabbit model, Eagle and co-workers in the 1940's found that established later stage disease required a longer course of therapy but cure could be accomplished with relatively low dose therapy (27) given over a prolonged period of time. These early studies have served as the basic tenants in our approach to syphilis therapy.
Penicillin, the first antibiotic developed, was the first known effective antibiotic for T. pallidum and remains the treatment of choice today (12, 30, 94). The maximally treponemicidal serum concentration of penicillin is 0.36 µg/ml which can kill the organism in 6 to 9 hours (27). However, a concentration as low as 0.005 µg/ml can clear T. pallidum from chancres if it is maintained for 48 to 96 hours (27). In the rabbit model, viable treponemes may persist in the lymph nodes even after early lesions have been cleared. T. pallidum can regenerate if the serum penicillin concentration falls to sub-inhibitory levels for 24-30 hours (54).
Resistance to penicillin has not yet been found among clinical isolates (119) but there is the potential for the acquisition of extrachromosomally mediated antibiotic resistance since at least one strain has been shown to contain plasmid DNA (88).
T. pallidum is also susceptible to virtually all other β-lactam antibiotics. Based on studies done with the time honored in vivo rabbit model, amoxicillin (82), ceftriaxone (66), ceftizoxime (65), cefmetazole (3) and cefetamet (30) have been shown to be curative.
Macrolide antibiotics are also able to inhibit T. pallidum but not as efficiently as the β-lactam antibiotics. When erythromycin was directly compared to penicillin G for the treatment of rabbit skin syphilomas, a single injection of penicillin decreased the motile T. pallidum count by more than 250-fold, whereas single doses of erythromycin did not decrease the count significantly (6). Only after multiple injections of erythromycin was the number of motile treponemes decreased in amounts similar to that of penicillin. Azithromycin, which has the advantage of once daily dosing, also has activity against T. pallidum but there have been multiple reports of treatment failure and resistance (17, 52, 56, 63, 69, 70, 77, 79, 83, 97, 114, 115, 126). Thus macrolides are relegated to second line antibiotic treatment status.
Chloramphenicol, an antibiotic no longer available in the USA, has failed to eradicate infection in the rabbit model but has been successful in treating syphilis in humans (54). Because it concentrates in the CNS, it has utility in treating neurosyphilis.
Spectinomycin (94), the quinolone class of antibiotics (117), and clindamycin (6) have negligible activity against T. pallidum and are contraindicated.
Combination Drugs
Combination therapy for syphilis is no more efficacious than single drug therapy. The addition of amoxicillin and probenecid for 10 days to 2.4 million units of penicillin achieves similar result as penicillin alone (100).
The following points are important to note before a discussion of antimicrobial therapy for syphilis. First, as in the case of many infections, a natural immune response can clear T. pallidum independent of antibiotic usage. On the other hand, in states of immune impairment, especially with cell-mediated deficits such as occurs in poorly controlled HIV/AIDS), even the most effective antibiotics may fail to eradicate T. pallidum (73). Hence, antibiotics augment clearance and subsequent cure but do not guarantee it. Furthermore, the pharmacology of the antibiotic preparation must be considered, specifically whether it reliably reaches the CNS in adequate amounts to inhibit T. pallidum because the CNS is invaded in up to 40% of patients (71). Unfortunately, many of the current therapies recommended for syphilis do not sufficiently penetrate into the CNS to best assure a cure of incubating neurosyphilis which typically does not appear for over 10 years making it difficult to be 100% sure that no residual replication competent spirochetes remain (25, 26, 80). Finally, the marker currently use to determine success of therapy, decrease in non-treponemal titers four-fold, is only indirect evidence of therapeutic success.
Penicillin is the best studied and still remains the recommended therapy for syphilis (118). Initial studies done in the rabbit model showed that the minimal inhibitory concentration that cures primary lesions is easily achieved with this dose of benzathine penicillin (54). The original studies of penicillin treatment in humans were performed before the establishment of the standard double-blinded placebo or comparison controlled randomized clinical trials to evaluate the efficacy of therapy. Since that time, there have been many observational and retrospective studies of therapy with penicillin with success rates, using a fourfold decrease in titers, which have varied from 75-100% (18). However, there are only a few randomized studies comparing penicillin to other antibiotics and only one study evaluating dosing schedules (1, 2, 18, 37, 98). Rigorous studies of penicillin to evaluate dose, duration, and preparation of antibiotic that would achieve cure with minimal toxicity and cost are severely lacking. After nearly 75 years, there still remains much debate as to what dosing schedules and which preparations are appropriate for the various stages and types of persons infected with T. pallidum (99). Hence, all recommendations of treatment for syphilis must be regarded with a degree of skepticism.
Because therapy should be tailored according to the benefit that a person will receive from a particular therapy balanced with the inconvenience and cost of alternative forms of therapy, the following discussion will focus on therapeutic options rather than rigorous recommendations.
ANTIMICROBIAL THERAPY
The following points are important to note before a discussion of antimicrobial therapy for syphilis. First, as in the case of many infections, a natural immune response can clear T. pallidum independent of antibiotic usage. On the other hand, in states of immune impairment, especially with cell-mediated deficits such as occurs in poorly controlled HIV/AIDS), even the most effective antibiotics may fail to eradicate T. pallidum (73). Hence, antibiotics augment clearance and subsequent cure but do not guarantee it. Furthermore, the pharmacology of the antibiotic preparation must be considered, specifically whether it reliably reaches the CNS in adequate amounts to inhibit T. pallidum because the CNS is invaded in up to 40% of patients (71). Unfortunately, many of the current therapies recommended for syphilis do not sufficiently penetrate into the CNS to best assure a cure of incubating neurosyphilis which typically does not appear for over 10 years making it difficult to be 100% sure that no residual replication competent spirochetes remain (25, 26, 80). Finally, the marker currently use to determine success of therapy, decrease in non-treponemal titers four-fold, is only indirect evidence of therapeutic success.
Penicillin is the best studied and still remains the recommended therapy for syphilis (118). Initial studies done in the rabbit model showed that the minimal inhibitory concentration that cures primary lesions is easily achieved with this dose of benzathine penicillin (54). The original studies of penicillin treatment in humans were performed before the establishment of the standard double-blinded placebo or comparison controlled randomized clinical trials to evaluate the efficacy of therapy. Since that time, there have been many observational and retrospective studies of therapy with penicillin with success rates, using a fourfold decrease in titers, which have varied from 75-100% (18). However, there are only a few randomized studies comparing penicillin to other antibiotics and only one study evaluating dosing schedules (1, 2, 18, 37, 98). Rigorous studies of penicillin to evaluate dose, duration, and preparation of antibiotic that would achieve cure with minimal toxicity and cost are severely lacking. After nearly 75 years, there still remains much debate as to what dosing schedules and which preparations are appropriate for the various stages and types of persons infected with T. pallidum (99). Hence, all recommendations of treatment for syphilis must be regarded with a degree of skepticism.
Because therapy should be tailored according to the benefit that a person will receive from a particular therapy balanced with the inconvenience and cost of alternative forms of therapy, the following discussion will focus on therapeutic options rather than rigorous recommendations.
2010 CDC Recommendations
The CDC periodically reviews the recommended treatments for syphilis (Table 3) (12). The CDC treatment guidelines are based on the premise that different stages or forms of the disease should be treated with different preparations and doses of penicillin. Following the principles of public health or population based guidelines; their goal is to provide therapeutic recommendations that will cure the greatest number of patients with minimal expense, easy administration, and high compliance (41). In contrast, treatment of the individual patient rests with the treating clinician who must weigh the risk of failing to cure a curable disease.
Because the CNS infection occurs in less than 40% of patients and the poor penetration of penicillin is inconsequential in the vast majority of immunocompetent hosts, penicillin and other antibiotics are capable of clearing the infection before a chronic infection or any neurological damage occurs, the CDC recommends benzathine penicillin, a penicillin which does not reliably achieve treponemicidal levels in the CNS, for early syphilis in all immunocompetent patients. Non-pregnant women and men with early syphilis, which includes primary syphilis (the stage that occurs immediately after infection characterized by an ulcer or chancre); secondary syphilis (the later stage corresponding to spirochete dissemination and characterized by skin rash); and early latent syphilis (the stage up to one year after initial infection when positive serology's are the only indication of infection), should be treated intramuscularly with one injection of 2.4 million units of benzathine penicillin (12).
The CDC also recommends that non-pregnant women and men with late latent syphilis (syphilis of more than 1-year in duration), latent syphilis of unknown duration, and tertiary syphilis except neurosyphilis) also receive benzathine penicillin (Table 2) but at an increased total dose of 7.2 million units given as 2.4 million units weekly for three weeks (12) to account for the possible longer dividing time of the spirochete at this stage of disease (54).
Acknowledging the well-recognized fact the neurosyphilis is the most difficult manifestation to treat, the CDC recommends that persons with neurosyphilis be treated with 3 - 4 million units of aqueous crystalline penicillin G given intravenously every 4 hours (18-24 million units daily) for 10-14 days. However, who should be included in this classification of "neurosyphilis" is vague and ill defined. Patients with a reactive cerebrospinal fluid VDRL or RPR test are the easiest to classify and are accepted as definitely having neurosyphilis. But most patients have either no symptoms or ill-defined symptoms and most have nonspecific CSF abnormalities. Furthermore, despite the fact that T. pallidum invades the CNS in 40% of patients (71), a lumbar puncture is seldom performed unless the signs and symptoms of acute meningitis are manifested as part of the signs and symptoms of secondary syphilis. In fact, the CDC discourages performing a lumbar puncture in early syphilis unless the patient has optic, auditory, cranial nerve, meningeal symptoms, or other indications of .possible neurosyphilis (12). The rationale is that although early invasion of the CNS by T. pallidum is common, it does not predict treatment failure with benzathine penicillin and that the lumbar puncture often yields little additive information.
Treatment Failures
Although benzathine penicillin is effective therapy for the vast majority of persons with syphilis, there are multiple reports of treatment failures, especially in patients co-infected with Human Immunodeficiency Virus (HIV). Although this failure can be at times attributed to re-infection (26, 31), many of those cases are convincingly due to failure of initial therapy (106). When a fourfold decrease in titers is the measure used to determine therapeutic success, re-treatment is necessary in at least 5% of immunocompetent patients with primary or secondary syphilis treated with low dose aqueous benzathine procaine penicillin and, if retreatment is required, it is only successful in 27% of patients(7, 99, 100, 106, 107, 108). The most likely reason for treatment failures is that at least 40% of patients with early syphilis are infected with T. pallidum in the CNS, a site that is poorly penetrated by benzathine penicillin (25, 26, 33, 61, 71, 80, 85, 95, 123, 128).
Even more concerning is that, even when titers fall, benzathine or low dose aqueous or procaine penicillin is not 100% successful in preventing neurosyphilis (24). Some patients, especially those with HIV, will go on to develop late neurosyphilis in 10-25 years. Unfortunately, the diagnosis is difficult to establish and penicillin therapy after the diagnosis of syphilis rarely reverses neurological deficits (54).
The clinician must weigh the risk of CNS invasion in each individual patient in deciding whether or not benzathine penicillin is adequate. This assessment should take into account immunocompetent of the host and stage of disease.
Most Effective Therapy for All Stages
Since the most reliable form of treatment is one that is reaches treponemicidal levels in the CNS, the most reliable therapy for any form of syphilis in any type of patient is 3 - 4 million units of aqueous crystalline penicillin G given intravenously (iv) every 4 hours (12-24 million units daily) for 10-14 days (Table 3) since between 5 and 24 million units of intravenous penicillin G consistently achieves levels in the CSF that are treponemicidal (80, 95). Hence, it would follow that treatment failures would be rare when compared to regimens that failed to reliably penetrate the CNS (84). This regimen is the most likely to prevent late neurological deficits which is rarely reversed even after treatment. However, it is rarely used even in immunocompromised patients except after neurological deficits have developed since inpatient hospital treatment is not always practical from a reimbursement or lifestyle perspective.
If outpatient therapy that achieved treponemicidal levels in the CNS is desired, 2.4 million units of aqueous procaine penicillin G intramuscularly daily with probenecid 500mg orally 4 times per day for 10-14 days can be given as an alternative therapy. Treponemicidal levels of penicillin have not been achieved as reliably with procaine penicillin but supplementation with probenecid will achieve treponemicidal levels (26). This option is uncommonly used since patient compliance is problematic because of the pain associated with repeated injections.
Alternative Therapy
The semi-synthetic penicillin, amoxicillin, when given with probenecid given 3.0 gm orally twice a day with probenecid 500 mg orally for 10-14 days is an alternative (Table 4). Probenecid inhibits renal excretion of natural and semi- synthetic penicillins. As previously noted, amoxicillin is treponemicidal in the rabbit model, and when 2 gm of amoxicillin plus 500mg of probenecid is administered to fasting adults, the MIC for T. pallidum can be exceeded for at least eight hours (29, 105). Clinical trials, albeit small, have confirmed the utility of this regimen (29, 90, 100).
The first- and third- generation cephalosporin antibiotics, in particular ceftriaxone, are also effective in treating syphilis. Ceftriaxone has been recommended as an alternative for treatment of early disease and "might be effective for treating late latent syphilis or syphilis of unknown duration" (Table 3) (12). This drug is particularly attractive since it readily crosses the blood brain barrier. Both serum and CSF levels exceeding those needed to kill T. pallidum are easily obtained with standard dosing (66). It has been used clinically to successfully to treat incubating syphilis (55), primary syphilis (55, 81, 96, 105, 113) , secondary syphilis (55, 96, 105), latent syphilis (24), and neurosyphilis (24, 53, 74). However, there have been reports of treatment failures in HIV infected patients, similar to those found with benzathine penicillin (24, 96, 111).
Doxycycline (100 mg twice daily) is also an effective alternative for treatment of immunocompetent adults with syphilis. In retrospective and observational studies, success rates vary from 73% to 100% using 10-14 days of therapy with doxycycline or tetracycline in early syphilis (26, 31, 39, 67, 91, 96, 106, 124). Although there is limited data on late latent syphilis, doxycycline is another alternative but the course of therapy should be extended to 28 days (91). The enthusiasm for this regimen is restrained because it is bacteriostatic in vitro and not bacteriocidal (119). Furthermore tetracycline and doxycycline are contraindicated in pregnant women and children under the age of 8 because of potential damage to developing bones and teeth (87).
Other regimens that are less desirable because of poor CSF penetration and reports of resistance are the macrolide antibiotics. Erythromycin had a failure rate of 14% after a follow up of 18 months (106). Another macrolide, azithromycin, has been shown in a recent meta-analysis to be more effective than benzathine penicillin G for treating early syphilis (1). However, in addition to its probable inadequate penetration into the CSF (109), it has been associated with resistance and treatment failures and therefore it can only be recommended when there are no other alternatives (17, 63, 70, 77, 79, 83, 97, 114, 115, 126).
Special Situations
Congenital Syphilis
For newborns, the treatment is 50,000 units/kg of aqueous crystalline penicillin G intravenously every 12 hours for a total of 100,000-150,000 units/kg/day for the first 7 days of life and then every 8 hours for a total of 10 days (12). The alternative regimen is 50,000 units/kg of procaine penicillin intramuscularly daily for 10 days. If treatment is interrupted for more than 24 hours, then the full course of therapy is restarted. Children diagnosed beyond the newborn period with congenital syphilis should also be treated presumptively for CNS involvement with therapy as described above except that aqueous crystalline penicillin needs to be administered every 4-6 hours instead of every 8 or 12 hours (110).
Optic Syphilis
Syphilitic involvement of the eye, a relatively common occurrence in HIV co-infected persons, should be treated with penicillin G 12 to 24 million units per day for 10 to 14 days regardless of CSF findings (21). Penicillin, however, regardless of dose or preparation, does not easily achieve treponemicidal levels in anterior chamber of the eye and hence, requires high doses and prolonged treatment (14 days) to improve the chances of adequate aqueous fluid levels (119). An illustrative case is that of a newborn with congenital syphilis receiving penicillin G potassium 50,000 units/kg body weight given intramuscularly at 12 hour intervals for 17 days after birth and T. pallidum were found in both the aqueous humor and ground eye tissue at autopsy (47). Hence, it may also be prudent to consider chloramphenicol or cephalothin, antibiotics that do achieve adequate levels in the anterior chamber of the eye, as possible first line agents (119). Because of excellent CNS penetration, chloramphenicol in a dose of 2 gm daily for 30 days is another alternative for optic syphilis.
Otosyphilis
As is the case of penetration into the anterior chamber of the eye, penicillin does not easily penetrate the labyrinth of the inner ear (119). Hence, a neurosyphilis regimen is warranted. Patients are usually treated with a prolonged course of penicillin for six weeks to three months with the addition of steroids (16, 23, 129). Oral prednisone 30-60 mg daily or every other day for at least 7 to 8 days should be given as tolerated by the patient (129). The utility of the steroids has never been proven but has become the standard of care to guard against the sudden worsening of hearing loss attendant to an acute reaction to released antigens form lysing organisms (Jarish-Herxheimer Reaction).
HIV Infected Patients
Although treatment failures and progression to syphilis appear to be less common in HIV infected patients on effective antiretroviral therapy (ART) (40, 41), immunosuppression associated with HIV infection and its deleterious effect on syphilis outcomes has been well described. More florid clinical manifestations, including multiple and more frequent genital ulcers (103) and symptomatic neurosyphilis, occurring early in the course of syphilis suggestive of a high spirochete burden (61), have been well documented in persons co-infected with HIV. In addition, multiple case reports suggest that HIV infected patients are more likely to fail antibiotic therapy than non-HIV infected patients (5, 61, 71, 86). For example, T. pallidum was isolated in three of three HIV infected patients evaluated after single dose benzathine penicillin therapy for early syphilis but could be cultured in the only patient without HIV infection (71). In addition, in a study of 59 patients with neurosyphilis, HIV infected patients were 2.5 and AIDS patients were 3.7 times less likely to normalize CSF-VDRL (75). Multiple retrospective chart reviews have documented treatment failures in up to 20% of HIV infected patients (24, 73), significantly higher than the 5-10% failure rate reported prior to the AIDS era. Furthermore, unlike many treatment failures in immunocompetent hosts, these failures are more likely to be associated with clinical disease, especially neurosyphilis (22, 24, 73).
More florid clinical manifestations and the higher frequency of treatment failures are most likely due to immune dysfunction coincident with HIV that results in a significantly higher spirochete load. There is a greater likelihood that spirochetes will remain active in sequestered sites. Hence, in HIV infected patients who are immunosuppressed and not on effective ART, it is prudent to consider use of antibiotics that more reliably penetrate the CNS. such as high dose penicillin or amoxicillin with probenecid.
Jarisch-Herxheimer reaction
Treatment for syphilis is not necessarily benign. Along with the discomfort of intramuscular or intravenous therapy and the possibility of a drug reaction, an acute febrile response to therapy is seen in 10-25% of all patients and 70-90% of patients with secondary syphilis (119). It is rarely seen in latent syphilis and occurs in varying amounts in the various forms of tertiary syphilis (104). This reaction, called the Jarisch-Herxheimer reaction, is most commonly associated with penicillin therapy but also has been associated with other antibiotics used to treat syphilis (4). It has been correlated with the release of heat stable "pyrogens" from T. pallidum and other spirochete infections (119) probably resulting in a rapid high release of inflammatory cytokines and chemokines. The clinical course is marked by the abrupt onset of symptoms usually beginning within two hours, occasionally as late as 24 hours, after initiation of therapy (12, 119). Besides fever, clinical signs and symptoms may include chills, headache, nausea, vomiting, myalgias, tachycardia, hyperventilation, mild hypotension, vasodilatation with flushing, and the exacerbation and new onset of skin lesions (104). It lasts 12-24 hours and is generally safe and self-limited (119). The exceptions are during the second half of pregnancy when there a risk of premature labor and fetal distress (12) and during cardiovascular, neurosyphilis and otosyphilis when reactions are more severe and can be life threatening (119). Acetaminophen or aspirin can be used for symptomatic relief. In severe cases or those associated with cardiovascular syphilis or neurosyphilis, prednisone 60 mg can be given. When given intravenously, this usually results in a prompt and dramatic decrease in fevers (119).
Penicillin Desensitization
Since penicillin is the optimal therapy for syphilis, penicillin should be seriously considered even in patients with penicillin allergies, except in those with a history of Stevens-Johnson syndrome, or alternatives are limited. These situations include syphilis in pregnant women, infants with congenital syphilis and adults with symptomatic neurosyphilis since treatment must be effective as soon as possible to prevent irreversible neurological disease. In addition, in syphilis in pregnancy and congenital syphilis, the toxicities associated with the tetracyclines make this option undesirable not only in terms of possible treatment failures but also in terms of damage to bone and teeth development (87).
Allergic skin reactions to penicillin are the principle toxicity associated with this antibiotic, are much less frequent with the pure preparations used today and occurred in at most 4% treatment courses in the past with less pure product (122). In addition, the vast majority of patients who report a history of penicillin allergy will not have an allergic reaction upon re-challenge.
Anaphylactic reactions are extremely rare today. The most reliable method of distinguishing those who will have a future reaction from those who will not is skin testing. Prior to skin testing, patients should be instructed not to take antihistamines, tricyclic antidepressants, and adrenergic drugs within a time interval appropriate for the corresponding half-life of the medication (12). For instance, chlorpheniramine maleate or terfenadine should not have been taken within 24 hours; diphenhydramine HCL or hydroxyzine within 4 days; and astemizole within 3 weeks. Testing is begun by injecting skin test reagents, including a negative diluent and positive histamine controls, in the epidermis on the volar surface of the forearm with a 26 gauge needle without drawing blood (122). If there is no reaction, then the same reagents are injected intradermally and if the diameter of induration is greater than 5 mm after 15 minutes, then the test is positive.
Optimally, skin testing should include diluents with both the major and minor determinants of penicillin (122). If the skin test is negative, then there is no risk of anaphylaxis but 1-3%t of patients will develop a mild cutaneous reaction. If the skin test is positive, then desensitization should be carried out prior to treatment since there is a 50-70% risk for an acute reaction. Because minor determinants of penicillin are not commercially available in the U.S., skin testing is often done with the major determinants only. In this case, if the test is negative then patients should still receive gradually increased doses of penicillin in monitored setting since there still is at least a 3% risk of a reaction.
Although desensitization can be done both orally and intravenously, oral desensitization is probably safer and certainly less expensive and easier (122). It should be performed in an intensive care setting with continuous electrocardiogram monitoring. Doses of penicillin should be gradually increased until the dose required for therapy is achieved. One-third of patients will develop a transient allergic reaction during desensitization but it is usually mild and self-limited. If mild symptoms occur, then the dose of penicillin should be stabilized until there are no signs or symptoms. If it is more sever, such as hypotension, laryngeal edema, or asthma, then the dose should be reduced at least ten-fold. There should be no interruption between desensitization and treatment since there the risk of an allergic reaction increases as the time from desensitization increases.
ENDPOINTS FOR MONITORING THERAPY
Non-HIV Infected Patients
As in any disease, the primary endpoint of therapy is clinical and a carefully taken medical history, physical exam and appropriate laboratory tests will determine if therapy has been successful. In primary syphilis, the patient should be examined for resolution of the chancre; in secondary syphilis, for resolution of the skin rash and any of the other protean disease manifestations that the patient may experience; and in early neurosyphilis for resolution of signs and symptoms of neurological disease. In late neurosyphilis, there is usually no reversal of signs and symptoms that are present prior to therapy but patients should be monitored carefully for further progression of disease. During any stage of syphilis and at any time, if there is persistence or recurrence of clinical disease, the person should be retreated. He or she should also be questioned about the possibility of re-infection to ease the concern about antibiotic resistance.
However, since the disease process is clinically silent at many stages and we are unable to grow in routine culture, it is necessary to follow disease activity with surrogate markers of infection. Treponemal antibody tests such as the FTA-abs are not helpful for measuring response to therapy since they are not quantified, usually remain positive for life, and do not correlate with disease activity (101, 112). In contrast, nontreponemal tests such as the RPR and VDRL are quantifiable, revert to negative in the vast majority of patients given effective therapy, and do correlate with disease activity. In fact, without therapy, 25% of patients eventually have non-reactive VDRL tests (48). With therapy, nearly 100% of non-HIV infected persons change from reactive to non-reactive.
Fiumara found that all 196 patients with treated primary syphilis had non-reactive RPR titers at one year (31) and all 204 patients with treated secondary syphilis by two years (32). In addition, he found that only 44% of 275 patients treated for late latent syphilis became seronegative (33). In another series, all patients with primary syphilis had resolution both clinically and serologically but only 55% of patients with secondary syphilis (84). However, some studies have shown that low but positive titer VDRL or RPR tests of 1:16 or less may persist despite no other evidence of treatment failure (the "serofast reaction") (12, 106). This may represent a false positive result or, especially when the titer is 1:4, persistent active infection or reinfection (119). HIV-infected patients are more likely to have a persistently high-titer RPR or VDRL.
Since VDRL titers are often slightly lower than RPR titers and there is significant variation in values between laboratories, either a RPR or VDRL, is best obtained from the same laboratory each time to monitor therapy (12). A fourfold change in titers, equivalent to a two dilution change, is considered a significant change and implies a change in disease activity. For instance, a change from 1:64 to 1:8 is significant while a change from 1:64 to 1:32 is not. The frequency of obtaining serological testing after treatment depends on the stage of disease and characteristics of the patients. Non-HIV infected adults with syphilis should be tested at three, six, and twelve months (119). If syphilis is greater than one year in duration or of unknown duration, then serologic testing should also be checked at 24 months. Pregnant women who are infected should have follow up serology in the third trimester and at delivery (12). If a pregnant woman is at a high risk for re-infection or in an area where the prevalence of syphilis is high, she should be retested every month (12). Infants with congenital syphilis and those who are sero-positive only because of passive transfer of maternal antibodies and are not infected should be checked with serological testing every two to three months (12).
The rate of decline after treatment of nontreponemal titers appears to be related to the stage of disease, height of the initial titers, and a prior history of syphilis (7, 31, 32, 33, 34). Brown and co-workers found a fourfold decrease in RPR titers at 6 months and eightfold decrease at 12 months in successfully treated patients with primary and secondary syphilis but there was a slower decline in patients with early latent disease and longer duration of symptoms (7). In contrast, patients with no prior history of syphilis (re-infection) have a more rapid decline in titers (7, 31, 32, 35). In addition, patients with macular rashes during the secondary stage revert to negative quicker than those with papular rashes (34). When titers do not decline fourfold by six months in primary or secondary syphilis or patients have a positive RPR/VDRL reaction beyond 12 months in primary syphilis, 24 months in secondary or latent syphilis, or 5 years in late syphilis, then patients should have a CSF examination and be retreated (94).
In adequately treated congenital syphilis, titers should decline by three months of age and should be non-reactive by six months of age (12). Titers, however, may decline slower if treatment is not initiated in the neonatal period and, occasionally, titers are still positive up to one year after birth. If titers are found to be stable or increasing or are still present after one year of age, then the child should be re-evaluated fully, including a CSF examination, and retreated.
Besides following serum nontreponemal test, a lumbar puncture is recommended every six months after treatment for neurosyphilis, including infants with abnormal CSF analyses, until the pleocytosis has resolved or CNS nontreponemal antibodies disappear (119). Pleocytosis usually resolves before CSF protein declines and CSF-VDRL tests may remain reactive for more than one year (61). If the CSF cell count is not decreased by six months or remains abnormal two years after therapy, re-treatment is indicated (8). In addition, in infants if the CSF-VDRL is still positive at six months of age, the infant should be retreated.
HIV Infected Patients
Although HIV infected patients may have similar non-treponemal (VDRL and RPR) response as non-HIV infected persons (43, 49), HIV infected patients are more prone to have a delayed serological response to infection or therapy and greater likelihood of having persistently reactive non-treponemal titers after treatment (91). Furthermore, HIV infected patients are more likely to have high non-treponemal antibody titers in secondary syphilis, e.g. 1:128 dilution (58), an increased rate of false positive non-treponemal antibody titers (47) and are less likely to have a fourfold decrease in titers within 6 months than matched non-HIV infected patients (97, 116). In addition, specific treponemal tests (FTA-ABS, TPHA and MHA-TP) more often become non-reactive in HIV infected patients than in non- HIV infected patients and, as would be expected, this loss of reactivity is associated with lower CD4 lymphocyte counts (46, 49).
This abnormal response to therapy is most likely due to immune dysfunction related to the increased polyclonal B cell activation found in HIV infection rather than a true change in disease activity, although the resultant loss of effective T-cell function could result in less ability to clear the spirochete (116).
VACCINES
No vaccines are currently available.
PREVENTION
Obviously, the most effective prophylaxis against contracting syphilis is avoidance of sexual contact with persons who harbor the spirochete. Condoms use during sexual intercourse can also be inferred to be protective from studies of HIV infection. Currently, prophylactic drugs are not recommendations in any group of patients with syphilis except those who have been exposed to a person with syphilis and in this population, it is unclear whether medicines prevent infection or treat very early disease, e.g. "incubating" syphilis.
We recommend that persons sexually exposed to primary, secondary, or latent syphilis of less than 1- year's duration within the previous 90 days be treated for syphilis even if they have negative serological tests for syphilis because there is data to suggest that therapy is most successful when initiated early and the treponemal burden is relatively low (47). Treatment with 2.4 million units of benzathine penicillin is recommended. Transmission occurs only when mucocutaneous lesions are present, which is usually within one year after initial infection (119), so contact with persons with syphilis greater than one year in duration is not an immediate indication for treatment (12). Instead, it is recommended that these persons be followed clinically and serologically for syphilis. Persons with exposure greater than 90 days prior to evaluation need not have immediate treatment, unless they have positive serological test, serological results will be delayed or follow up is uncertain.
Blood transmission is rare today because of the low incidence of disease and blood storage procedures.
REFERENCE
1 Bai ZG,et al. Azithromycin vs. benzathine penicillin G for early syphilis: a meta-analysis of randomized clinical trials. Int J Std AIDS. 2008;19:217-21.[PubMed]
2. Bai ZG, Yang KH, Liu YL, Tian JH, Ma B, Mi DH, Jiang L, Tan JY, Gai QY. Azithromycin vs. benzathine penicillin G for early syphilis. Cochrane Database Syst Rev. 2012;6:CD007270.
3. Baker-Zander SA, Lukehart SA. Efficacy of cefmetazole in the treatment of active syphilis in the rabbit model. Antimicrob Agents and Chemother. 1989;33:1465-9.[PubMed]
4. Beall GN. Penicillins. In: Saxon A, moderator. Immediate hypersensitivity reactions to beta-lactam antibiotics. Ann Intern Med 1987;107:204-15. [PubMed]
5. Berry CD, Hooton TM, Collier AC, Lukehart SA. Neurological relapse after benzathine penicillin therapy for secondary syphilis in a patient with HIV infection. N Engl J Med. 1987;316:1587-9.[PubMed]
6. Brause BD, Borges JS, Roberts RB. Relative efficacy of clindamycin, erythromycin, and penicillin in treatment of Treponema pallidum in skin syphilomas of rabbits. J Infect Dis. 1976;134:93-6.[PubMed]
7. Brown ST, Zaidi A, Larsen SA, Reynolds GH. Serological response to syphilis treatment: a new analysis of old data. JAMA. 1985;253:1296-9.[PubMed]
8. Castro R, Prieto ES, Santo I, Azevedo J, Exposto Fda L. Evaluation of an enzyme immunoassay technique for detection of antibodies against Treponema pallidum. J Clin Microbiol. 2003 ;41:250-3.[PubMed]
9. Centers for Disease Control and Prevention. Infectious Diseases Laboratories. Test Order: Treponema pallidum Molecular Detection. [2013 January 22]. Retrieved from http://www.cdc.gov/laboratory/specimen-submission/detail.html?CDCTestCode=CDC-10176.
10. Centers for Disease Control and Prevention. The National Plan to Eliminate Syphilis from the United States. [2012 December]. Retrieved http://www.cdc.gov/std/stats11/surv2011.pdf
11. Centers for Disease Control and Prevention. Sexually Transmitted Disease Surveillance. [2010 December 17], Retrieved from http://www.cdc.gov/std/treatment/2010/.
12. Centers for Disease Control and Prevention. Sexually Transmitted Treatment Guidelines 2010. MMWR. 2010;59 (No.RR-12):1-110. [PubMed]
13. Centers for Disease Control and Prevention. Summary of Notifiable Diseases---United States, 2000.MMWR.2002;49):1-102.[PubMed]
14. Centers for Disease Control and Prevention. Syphilis Testing Algorithms Using Treponemal Tests for Initial Screening --- Four Laboratories, New York City, 2005—2006. MMWR 2008;57(32):872-5.[PubMed]
15. Centers for Disease Control and Prevention. Patton ME, Su JR, Nelson R, Weinstock H. Primary and Secondary Syphilis — United States, 2005–2013. MMWR 2014;63(18):402-6. [PubMed]
6. Chan YM, Adams A, Ker AG. Syphilitic labyrinthitis - an update. J Laryngol Otol. 1995;109:719-25.[PubMed]
17. Chen XS, Yin YP, Wei WH, Wang HC, Peng RR, Zheng HP, Zhang JP, Zhu BY, Liu QZ, Huang SJ. High prevalence of azithromycin resistance to Treponema pallidum in geographically different areas in China. Clin Microbiol Infect. 2013;19:975-9. [PubMed]
18. Clement ME, Okeke L, Hicks, CB. Treatment of Syphilis: A Systematic Review. JAMA. 2014;312:1905-17. [PubMed]
19. Cummings MC, Lukehart SA, Marra C, Smith BL, Shaffer J, Demeo LR, Castro C, McCormack WM. Comparison of methods for the detection of Treponema pallidum in lesions of early syphilis. Sex Transm Dis. 1996;23:366-9.[PubMed]
20. Diagnostics Direct, LLC. 510(k) SUMMARY [2011-Aug]. Retrieved from http://www.accessdata.fda.gov/cdrh_docs/pdf10/K102400.pdf .
21. Deschenes J, Seamone CD, Baines MG. Acquired ocular syphilis: diagnosis and treatment. Ann Ophthalmol. 1992;24:134-8.[PubMed]
22. Dibbern DA Jr, Ray SC. Recrudescence of treated neurosyphilis in a patient with human immunodeficiency virus. Mayo Clin Proc. 1999;74:53-6.[PubMed]
23. Dobbin JM, Perkins JH. Otosyphilis and hearing loss: response to penicillin and steroid therapy. Laryngoscope. 1983;93:1540-2.[PubMed]
24. Dowell ME, Ross PG, Musher DM, Cate TR, Baughn RE. Response of latent syphilis or neurosyphilis to ceftriaxone therapy in persons infected with human immunodeficiency virus. Am J Med. 1992;93:481-8.[PubMed]
25. Ducas J, Robson HG. Cerebrospinal fluid levels during therapy for latent syphilis. JAMA.1981;246:2583-4.[PubMed]
26. Dunlop EMC, Al-Egaily SS, Houang MB. Penicillin levels in blood and CSF achieved by treatment of syphilis. JAMA. 1979;241:2538-40. [PubMed]
27. Eagle H, Fleischman R, Magnusson AJ. The effective concentrations of penicillin in vitro and in vivo for streptococci, pneumococci, and Treponema pallidum. J Bacteriol. 1950;59:625-43. [PubMed]
28. Engelkens HJ, ten Kate FJ, Judanarso J, Vuzevski VD, van Lier JB, Godschalk JC, van der Sluis JJ, Stolz E. The localisation of treponemes and characterisation of the inflammatory infiltrate in skin biopsies from patients with primary or secondary syphilis, or early infectious yaws. Genitourin Med. 1993;69:102-7.[PubMed]
29. Faber WR, Bos JD, Rietra PJ, Fass H, Van Eijk RV. Treponemicidal levels of amoxicillin in cerebrospinal fluid after oral administration. Sexually Transm Dis. 1983;10:148-50.[PubMed]
30. Fitzgerald TJ. Effects of Cefetamet (Ro 15-8074) on Treponema pallidum and Experimental Syphilis. Antimicrob Agents and Chemother. 1992;36:598-602.[PubMed]]
31. Fiumara NJ. Treatment of seropositive primary syphilis: an evaluation of 196 patients. Sexually Transm Dis. 1977;4:92-5.[PubMed]32. Fiumara NJ. Treatment of secondary syphilis: an evaluation of 204 patients. Sexually Transm Dis. 1977;4:96-9.[PubMed]
33. Fiumara NJ. Serological responses to treatment of 128 patients with late latent syphilis. Sexually Transm Dis. 1979;6:243-6.[PubMed]
34. Fiumara NJ. Reinfection primary, secondary, and latent syphilis: the serological response after treatment. Sex Trans Dis. 1980;7:111-5.[PubMed]
35. Fiumara NJ. Treatment of primary and secondary syphilis. Serological response. JAMA. 1980;243:2500-2.[PubMed]
36. Fraser CM, Norris SJ, Weinstock GM, White O, Sutton GG, Dodson R, Gwinn M, Hickey EK, Clayton R, Ketchum KA, Sodergren E, Hardham JM, McLeod MP, Salzberg S, Peterson J, Khalak H, Richardson D, Howell JK, Chidambaram M, Utterback T, McDonald L, Artiach P, Bowman C, Cotton MD, Fujii C, Garland S, Hatch B, Horst K, Roberts K, Sandusky M, Weidman J, Smith HO, Venter JC. Complete genome sequence of Treponema pallidum, the syphilis spirochete. Science. 1998;281:375-88.[PubMed]
37. Ganesan A, Mesner O, Okulicz JF, O'Bryan T, Deiss RG, Lalani T, Whitman TJ, Weintrob AC, Macalino G, Agan BK. A single dose of benzathine penicillin G is as effective as multiple doses of benzathine penicillin G for the treatment of HIV-infected persons with early syphilis. Clin Infect Dis. 2014; Nov 10. pii: ciu888. [Epub ahead of print.] [PubMed]
38. Genest DR, Choi-Hong SR, Tate JE, Qureshi F, Jacques SM, Crum C. Diagnosis of congenital syphilis from placental examination: comparison of histopathology, Steiner stain, and polymerase chain reaction for Treponema pallidum DNA. Hum Pathol. 1996;27:366-72. [PubMed]
39. Ghanem KG, Erbelding EJ, Chen WW, Rompalo AM. Doxycycline compared with benzathine penicillin for the treatment of early syphilis. Clin Infect Dis. 2006;42:e45-9. [PubMed]
40. Ghanem KG, Moore RD, Rompalo AM, Erbelding EJ, Zenilman JM, Gebo KA. Antiretroviral therapy is associated with reduced serologic failure rates for syphilis among HIV-infected patients. Clin Infect Dis. 2008;47:258-65. [PubMed]
41. Ghanem KG, Moore RD, Rompalo AM, Erbelding EJ, Zenilman JM, Gebo KA. Neurosyphilis in a clinical cohort of HIV-1-infected patients AIDS. 2008;22:1145-51. [PubMed]
42. Gottlieb SL, Pope V, Sternberg MR, McQuillan GM, Beltrami JF, Berman SM, Markowitz LE. Prevalence of syphilis seroreactivity in the United States: data from the National Health and Nutrition Examination Surveys (NHANES) 2001-2001. Sex Transm Dis. 2008;35:507-11. [PubMed]
43. Gourevitch MN, Selwyn PA, Davenny K, Buono D, Schoenbaum EE, Klein RS, Friedland, GH. Effects of HIV infection on the serological manifestations and response to treatment of syphilis in intravenous drug users. Ann Intern Med .1993;118:350-5.[PubMed]
44. Grange PA, Gressier L, Dion PL, Farhi D, Benhaddou N, Gerhardt P, Morini JP, Deleuze J, Pantoja C, Bianchi A, Lassau F, Avril MF, Janier M, Dupin N. Evaluation of a PCR test for detection of treponema pallidum in swabs and blood. J Clin Microbiol. 2012;50:546-52. [PubMed]
45. Grimprel E, Sanchez PJ, Wendel GD, Burstain JM, McCracken GH, Radolf JD, Norgard MV. Use of polymerase chain reaction and rabbit infectivity testing to detect Treponema pallidum in amniotic fluid, fetal and neonatal sera, and cerebrospinal fluid. J Clin Microbiol. 1991;29:1711-8.[PubMed]
46. Haas JS, Bolan G, Larsen SA, Clement MJ, Bacchetti P, Moss AR. Sensitivity of treponemal tests for detecting prior treated syphilis during human immunodeficiency virus infection. J Infect Dis. 1990;162:862-6.[PubMed]
47. Hardy JB, Hardy PH, Oppenheimer EH, Ryan SJ, Sheff RN. Failure of penicillin in a newborn with congenital syphilis. JAMA. 1970;212:1345-9.[PubMed]
48. Hart G. Syphilis tests in diagnostic and therapeutic decision making. Ann Intern Med. 1986;104:368-76.[PubMed]
49. Hazlett KR, Cox DL, Decaffmeyer M, Bennett MP, Desrosiers DC, La Vake CJ, La Vake ME, Bourell KW, Robinson EJ, Brasseur R, Radolf JD. TP0453, a concealed outer membrane protein of Treponema pallidum, enhances membrane permeability. J Bacteriol. 2005;187:6499-508.[PubMed]
50. Herring A, Ballard R, Mabey D, Peeling RW; WHO/TDR Sexually Transmitted Diseases Diagnostics Initiative. Evaluation of rapid diagnostic tests: syphilis. Nat Rev Microbiol. 2006; 4(12 Suppl):S33-40.[PubMed]
51. Heymans R, van der Helm JJ, de Vries HJ, Fennema HS, Coutinho RA, Bruisten SM. Clinical value of Treponema pallidum real-time PCR for diagnosis of syphilis. J Clin Microbiol. 2010;48:497-502. [PubMed]
52. Hook EW, Stephens J, Ennis DM. Azithromycin compared with penicillin G benzathine for treatment of incubating syphilis. Ann Intern Med. 1999;131:434-7.[PubMed]
53. Hook EW, Baker-Zander SA, Moskovitz BL, Lukehart SA, Handsfield HH. Ceftriaxone therapy for asymptomatic neurosyphilis - case report and western blot analysis of serum and cerebrospinal fluid IgG response to therapy. Sex Transm Dis. 1986;13:185-8.[PubMed]
54. Hook EW, Marra CM. Acquired syphilis in adults. N Engl J Med. 1992;326:1060-9.[PubMed]
55. Hook EW, Roddy RE, Handsfield HH. Ceftriaxone for incubating and early syphilis. J Infect Dis. 1988;158:881-4.[PubMed]
56. Hook EW, Behets F, Van Damme K, Ravelomanana N, Leone P, Sena AC, Martin D, Langley C, McNeil L, Wolff M. A phase III equivalence trial of azithromycin versus benzathine penicillin for treatment of early syphilis. J Infect Dis. 2010;201:1729-35. [PubMed]
57. Hagedorn HJ, Kraminer-Hagedorn A, De Bosschere K, Hulstaert F, Pottel H, Zrein M. Evaluation of INNO-LIA syphilis assay as a confirmatory test for syphilis. J Clin Microbiol 2002;40:973-8.[PubMed]
58. Hutchinson CM, Hook EW, Shepard M, Verley J, Rompalo AM. Altered clinical presentation of early syphilis in patients with human immunodeficiency virus infection. Ann Intern Med. 1994;121:94-9.[PubMed]
59. Janier M, Chastang C, Spindler E, Strazzi S, Rabian C, Marcelli A, Morel P. A prospective study of the influence of HIV status on the seroreversion of serological tests for syphilis. Dermatology. 1999;198:362-9.[PubMed]
60. Jethwa HS, Schmitz JL, Dallabetta G, Behets F, Hoffman I, Hamilton H, Lule G, Cohen M, Folds JD. Comparison of molecular and microscopic techniques for detection of Treponema pallidum in genital ulcers. J Clin Microbiol. 1995;33:180-3.[PubMed]
61. Johns DR, Tierney M, Felsenstein D. Alteration in the natural history of neurosyphilis by concurrent infection with the human immunodeficiency virus. N Engl J Med. 1987;316:1569-72.[PubMed]
62. Joyanes P, Borobio MV, Arquez JM, Perea EJ. The association of false-positive rapid plasma reagin results and HIV infection. Sex Transm Dis. 1998;25:569-71.[PubMed]
63. Katz KA, Klausner JD. Azithromycin resistance in Treponema pallidum. Curr Opin Infect Dis. 2008;21:83-91.[PubMed]
64. Koek AG, Bruisten SM, Dierdorp M, vab Dam AP, Templeton K. Specific and sensitive diagnosis of syphilis using a real-time PCR for Treponema pallidum. Clin Microbio Infect 2006;12:1233-1236. [PubMed]
65. Korting HC, Haag R, Walter D, Riethmuller U, Meurer M. Efficacy of ceftizoxime in the treatment of incubating syphilis in rabbits. Chemotherapy. 1993;39:331-5.[PubMed]
66. Korting HC, Walther D, Riethmuller U, Meurer M. Comparative in vitro susceptibility of Treponema pallidum to ceftizoxime, ceftriaxone, and penicillin G. Chemotherapy. 1986;32:352-5.[PubMed]
67. Li J, Zheng HY. Early syphilis – serological treatment response to doxycycline/tetracycline vs benzathine penicillin. J Infect Dev Ctrie. 2014;8:228-32. [PubMed]
68. Liu H, Rodes B, Chen CY, Steiner B. New tests for syphilis: rational design of a PCR method for detection of Treponema pallidum in clinical specimens using unique regions of the DNA polymerase I gene. J Clin Microbiol. 2001;39:1941-6.[PubMed]
69. Lukehart SA, Fohn MJ, baker-Zander SA. Efficacy of azithromycin for therapy of active syphilis in the rabbit model. J Antimicrob Chemother. 1990;25:91-9.[PubMed]
70. Lukehart SA, Godornes C, Molini BJ, Sonnett P, Hopkins S, D., Mulcahy F, Engelman J, Mitchell SJ, Rompalo AM, Marra CM, Klausner JD. Macrolide resistance in Treponema pallidum in the United States and Ireland. N Engl J Med. 2004;351:154–8. [PubMed]
71. Lukehart SA, Hook EW III, Baker-Zander SA, Collier AC, Critchlow CW, Handsfield HH. Invasion of the central nervous system by Treponema pallidum: implications for diagnosis and treatment. Ann Intern Med .1988;109:855-62.[PubMed]
72. Lusiak M, Podwińska J. Interleukin 10 and its role in the regulation of the cell-mediated immune response in syphilis. Arch Immunol Ther Exp (Warsz). 2001;49:417-21.[PubMed]
73. Malone JL, Wallace MR, Hendrick BB, LaRocco A Jr, Tonon E, Brodine SK, Bowler WA, Lavin BS, Hawkins RE, Oldfield EC 3rd. Syphilis and neurosyphilis in a human immunodeficiency virus type-1 seropositive population: evidence for frequent serologic relapse after therapy. Am J Med. 1995;99:55-63.[PubMed]
74. Marra CM, Boutin P, McArthur JC, Hurwitz S, Simpson G, Haslett PA, van der Horst C, Nevin T, Hook EW 3rd. A pilot study evaluating ceftriaxone and penicillin G as treatment agents for neurosyphilis in human immunodeficiency virus-infected individuals. Clin Infect Dis. 2000;30:540-4.[PubMed]
75. Marra CM, Maxwell CL, Tantalo L, Eaton M, Rompalo AM, Raines C, Stoner BP, Corbett JJ, Augenbraun M, Zajackowski M, Kee R, Lukehart SA. Normalization of cerebrospinal fluid abnormalities after neurosyphilis therapy: does HIV status matter? Clin Infect Dis. 2004;38:1001-6.[PubMed]
76. Marra CM, Tantalo LC, Maxwell CL, Dougherty K, Wood B. Alternative cerebrospinal fluid tests to diagnose neurosyphilis in HIV-infected individuals. Neurology. 2004;63:85-8. [PubMed]
77. Martin IE, Tsang RS, Sutherland K, Anderson B, Read R, Roy C, Yanow S, Fonseca K, White W, Kandola K, Kouadjo E, Singh AE. Molecular typing of Treponema pallidum strains in western Canada: predominance of 14d subtypes. Sex Transm Dis. 2010;37:544-8. [PubMed]
78.Michelow IC, Wendel GD, Norgard MV, Zeray F, Leos NK, Alsaadi R, Sanchez PJ. Central nervous system infections in congenital syphilis. N Engl J Med. 2002;346:1792-8.[PubMed]
79. Mitchell SJ, Engelman J, Kent CK, Lukehart SA, Godornes C, Klausner JD. Azithromycin-resistant syphilis infection: San Francisco, California, 2000–2004. Clin Infect Dis. 2006;42:337–45.< [PubMed]
80. Mohr JA, Griffiths W, Jackson R, Saadah H, Bird P, Riddle J. Neurosyphilis and penicillin levels in cerebrospinal fluid. N Engl J Med. 1976;316:1569-72.[PubMed]
81. Moorthy TT, Lee C-T, Lim K-B, Tan T. Ceftriaxone for treatment of primary syphilis in men: a preliminary study. Sex Transm Dis. 1987;14:116-8.[PubMed]
82. Morrison RE, Harrison SM, and Tramont EC. Oral amoxycillin, an alternative treatment for neurosyphilis. Genitourin Med. 1985;61:359-62. [PubMed]
83. Muldoon EG, Walsh A, Crowley B, Mulcahy F. Treponema pallidum azithromycin resistance in Dublin, Ireland Sex Transm Dis. 2012;39:784-6. [PubMed]
84. Musher DM. How much penicillin cures early syphilis. Ann Intern Med. 1988;109:849-51.[PubMed]
85. Musher DM, Baughan RE. Neurosyphilis in HIV-infected persons. N Engl J Med. 1994;1516-7.[PubMed]
86. Musher DM, Hamill RJ, Baughn RE. Effect of human immunodeficiency virus (HIV) infection on the course of syphilis and on the response to treatment. Ann Intern Med. 1990;113:872-81.[PubMed]
87. Meyers B, Salvatore M. Tetracyclines and chloramphenicol. In: Mandell GL, Bennett JE, Dolin R. Principles and Practice of Infectious Diseases. 6th ed. New York: Churchill Livingston 2005:356-373.
88. Norgard MV, Miller JN. Plasmid DNA in Treponema pallidum (Nichols): potential for antibiotic resistance by syphilis bacteria. Science. 1981;213:553-5.[PubMed]
89. Norris SJ, Cox DL, Weinstock GM. Biology of Treponema pallidum: correlation of functional activities with gernome sequence data. J Mol Microbiol Biotechnol. 2001;3:37-62.[PubMed]
90. Onoda Y. Clinical evaluation of amoxycillin in the treatment of syphilis. J Int Med Res. 1979;7:539-45.[PubMed]
91. Onoda Y. Therapeutic effect of oral doxycycline on syphilis. Br J Vener Dis. 1979;55:110-5.[PubMed]
92. Palmer HM, Higgins SP, Herring AJ, Kingston MA. Use of PCR in the diagnosis of early syphilis in the United Kingdom. Sex Transm Infect. 2003 ;79 :479-53.[PubMed]
93. Peeling RW, Hook EW 3rd. The pathogenesis of syphilis: the great mimicker, revisitd. J Pathol 2006;208:224-32.[PubMed]
94. Peterman TA, Zaidi AA, Lieb S, Wroten JE. Incubating syphilis in patients treated for gonorrhea: a comparison of treatment regimens. J Infect Dis. 1994;170:689-92.[PubMed]
95. Polnikorn N, Witoonpanih W, Vorachit M, Vejjajiva S, Vejjajiva A. Penicillin concentrations in cerebrospinal fluid after different treatment regimens for syphilis. Br J Vener Dis. 1980;56:363-7.[PubMed]
96. Psomas KC, Brun M, Causse A, Atoui N, Reynes J, Le Moing V. Efficacy of ceftriaxone and doxycycline in the treatment of early syphilis. Med Mal Infect. 2012;42:15-9. [PubMed]
97. Read P, Jeoffreys N, Tagg K, Guy RJ, Gilbert GL, Donovan B. Azithromycin-resistant syphilis-causing strains in Sydney, Australia: prevalence and risk factors. J Clin Microbiol. 2014;52:2776-81. [PubMed]
98. Riedner G, Rusizoka M, Todd J, Maboko L, Hoelscher M, Mmbando D, Samky E, Lyamuya E, Mabey D, Grosskurth H, Hayes RN. Single-dose azithromycin versus penicillin G benzathine for the treatment of early syphilis. N Engl J Med. 2005;353:1236-44. [PubMed]
99. Rolfs RT. Treatment of syphilis, 1995. Clin Infect Dis. 1995;20:S23-38.[PubMed]
100. Rolfs RT, Joesoef MR, Hendershot EF, Rompalo AM, Augenbraun MH, Chiu M, Bolan G, Johnson SC, French P, Steen E, Radolf JD, Larsen S. A randomized trial of enhanced therapy for early syphilis in patients with and without human immunodeficiency virus infection. N Engl J Med. 1997;337:307-14.[PubMed]
101. Romanowski B, Sutherland R, Fick GH, Mooney D, Love EJ. Serologic response to treatment of infectious syphilis. Ann Intern Med. 1991;114:1005-9.[PubMed]
102. Rompalo AM, Joesoef MR, O'Donnell JA, Augenbraun M, Brady W, Radolf JD, Johnson R, Rolfs RT; Syphilis and HIV Study Group. Clinical manifestations of early syphilis by HIV status and gender: results of the syphilis and HIV study. Sex Transm Dis. 2001;28:158-65.[PubMed]
103. Rompalo AM, Lawlor J, Seaman P, Quinn TC, Zenilman JM, Hook EW. Modification of syphilitic genital ulcer manifestations by coexistent HIV infection. Sex Transm Dis. 2001;28:448-54.[PubMed]
104. Rosen T, Rubin H, Ellner K, Tshen J, Cochran R. Vesicular Jarisch-Herxheimer Reaction. Arch Dermatol. 1989;125:77-81.[PubMed]
105. Schofer H, Vogt HJ, Milbradt R. Ceftriaxone for the treatment of primary and secondary syphilis. Chemotherapy. 1989;35:140-5. [PubMed]
106. Schroeter AL, Lucas JB, Price EV, Falcone VH. Treatment for early syphilis and reactivity of serological tests. JAMA. 1972;221:471-6. [PubMed]
107. Seña AC, Wolff M, Behets F, Van Damme K, Martin DH, Leone P, McNeil L, Hook EW. Response to therapy following retreatment of serofast early syphilis patients with benzathine penicillin. Clin Infect Dis. 2013;56:420-2. [PubMed]
108. Short DH, Knox JM, Glicksman J. Neurosyphilis, the search for adequate treatment. Arch Dermat. 1966;93:87-91.[PubMed]
109. Jaruratanasirikul S, Hortiwakul R, Tantisarasart T, Phuenpathom N, Tussanasunthornwong S. Distribution of azithromycin into brain tissue, cerebrospinal fluid, and aqueous humor of the eye. Antimicrob Agents Chemother. 1996; 40: 825–6. [PubMed]
110. Saloojee H, Velaphi S, Goga Y, Afadapa N, Steen R, Lincetto O. The prevention and management of congenital syphilis: an overview and recommendations. Bull World Health Organ. 2004 Jun;82:424-30.[PubMed]
111. Smith NH, Musher DM, Huang DB, Rodriguez PS, Dowell ME, Ace W, White AC Jr. Response of HIV-infected patients with asymptomatic syphilis to intensive intramuscular therapy with ceftriaxone or procaine penicillin. Int J STD AIDS. 2004;15:328-32. [PubMed]
112. Sparling PF. Diagnosis and treatment of syphilis. N Engl J Med. 1971;284:642-53. [PubMed]
113. Spornraft-Ragaller P, Abraham S, Lueck C, Meurer M. Response of HIV-infected patients with syphilis to therapy with penicillin or intravenous ceftriaxone. Eur J Med Res. 2011;16:47-51. [PubMed]114. Stamm LV, Parish EA. In vitro activity of azithromycin and CP-63,956 against Treponema pallidum. J Antimicrob Chemother. 1990;25:11-4.[PubMed]
115. Su JR, Pillay A, Hook EW, Ghanem KG, Wong W, Jackson D, Smith LD, Pierce E, Philip SS, Wilson S, Golden MR, Workowski KA, Chi KH, Parrish DD, Chen CY, Weinstock HS. Prevalence of the 23S rRNA A2058G point mutation and molecular subtypes in Treponema pallidum in the United States, 2007 to 2009. Sex Transm Dis. 2012;39:794-8. [PubMed]
116. Telzak EE, Greenberg MSZ, Harrison J, Stoneburner RL, Schultz S. Syphilis treatment response in HIV-infected individuals. AIDS. 1991;5:591-5.[PubMed]
117. Tartaglione TA, Hooton TM. The role of fluoroquinolones in sexually transmitted disease. Pharmocotherapy.1993;13:189-201.[PubMed]
118. Tramont EC. Syphilis in adults: from Christopher Columbus to Sir Alexander Fleming to AIDS. Clin Infect Dis. 1995;21:1361-71.[PubMed]
119. Tramont EC. Treponema pallidum (syphilis). In: Mandell GL, Bennett JE, Dolin R. Principles and Practice of Infectious Diseases. 6th ed. New York: Churchill Livingston 2005:2768-85.
120. U.S. Food and Drug Administration. FDA grants CLIA waiver expanding the availability of rapid screening test for syphilis. [2014 December 15]. Retrieved from http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm426843.htm.
121. U.S. Preventive Services Task Force. Screening for Syphilis: Brief Update. July 2004. Agency for Healthcare Research and Quality, Rockville, MD.http://www.ahrq.gov/clinic/3rduspstf/syphilis/syphilup.htm
122. Weiss ME, Adkinson NF. β-lactam allergy. In: Mandell GL, Bennett JE, Dolin R. Principles and Practice of Infectious Diseases. 6th ed. New York: Churchill Livingston 2005:318-325.
123. Whiteside CM. Persistence of neurosyphilis despite multiple treatment regimens. Am J Med. 1989;87:225-7.[PubMed]
124. Wong T, Singh AE, De P. Primary syphilis: serological treatment response to doxycycline/tetracycline versus benzathine penicillin. Am J Med. 2008;121:903-8. [PubMed]
125. World Health Organization. Global incidence and prevalence of selected curable sexually transmitted intections – 2008. . 2012. http://apps.who.int/iris/bitstream/10665/75181/1/9789241503839_eng.pdf?ua=1
126. Wu BR, Yang CJ, Tsai MS, Lee KY, Lee NY, Huang WC, Wu H, Lee CH, Chen TC, Ko WC, Lin HH, Lu PL, Chen YH, Liu WC, Yang SP, Wu PY, Su YC, Hung CC, Chang SY. Multicentre surveillance of prevalence of the 23S rRNA A2058G and A2059G point mutations and molecular subtypes of Treponema pallidum in Taiwan, 2009-2013. Clin Microbiol Infect. 2014;20:802-7. [PubMed]
127. Yinnon AM, Coury-Doniger P, Polito R, Reichman RC. Serological response to treatment of syphilis in patients with HIV infection. Arch Intern Med. 1996;156:321-5.[PubMed]
128. Yoder FW. Penicillin treatment of neurosyphilis. Are recommended dosages sufficient? JAMA. 1975;232:270-1.[PubMed]
129. Zoller M, Wilson WR, Nadol JB. Treatment of syphilitic hearing loss. Ann Otol Rhinol Laryngol. 1979;88:160-5.[PubMed]
Tables
Table 1. Diagnostic Tests
| Test | Indication |
|---|---|
| Darkfield microscopy | Diagnosis of syphilis when accessible lesions such as chancres are present |
| Nontreponemal (reagenic) tests VDRL, RPR, ART, TRUST | Nonspecific first line screening test which requires a more specific confirmatory test. Titers determine whether or not a previously treated person needs retreatment |
| FTA-ABS | Confirmatory test. Test in primary and tertiary syphilis if there is a high index of suspicion if with a negative nontreponemal test |
| TP-PA, MHATP | Similar indications as FTA-ABS but easier to perform and not as sensitive, especially in early disease |
| ELISA | Screening test but not quantitative so nonspecific test required for confirmation and assessment of need for treatment |
| RELISPOT | Detects immunoglobulin-secreting cells and is a nonspecific indicator of infection. Most often used to diagnose congenital syphilis. |
| ICS | Point-of-care testing in developing countries |
| ICT | Immunochromatographic Membrane Test |
| LIA | Point-of-care testing in developing countries |
| Immunofluorescent and Immunoperoxidase Antibody Staining | Used to diagnose congenital syphilis |
| PCR | Not commercially available. May be used to diagnose congenital syphilis |
| Immunoblotting/Western blot | Used to diagnose congenital syphilis |
Table 2. Traditional Serological Tests (Modified from reference 92)
| Type of Test | Untreated Patients with Positive Tests (%) | ||
|---|---|---|---|
| Early (Primary, Secondary, and Early Latent) | Late (Late latent and Tertiary) | ||
| Nontreponemal (reaginic) VDRL, RPR, ART, TRUST | 70-100 | 60-98 | |
| Specific Treponemal FTA-abs, TPHA, MHA-TP, ELISA | 50-85 | 97-100 | |
Table 3. 2010 CDC Guidelines for Syphilis Treatment
| Stage | Patients not allergic to penicillin | Allergic to penicillin |
|---|---|---|
Primary, secondary, and early Latent For early syphilis, serum concentrations must exceed 0.018 ug/ml (0.03 unit per milliliter) for 7-10 days |
Adults: Benzathine penicillin G 2.4 million units im x 1 once Children: benzathine penicillin G 50,00 units/kg im up to adult dose of 2.4 million units once |
Nonpregnant: doxycycline 100 mg po bid or tetracycline 500 mg po qid x 14 days. If intolerant to doxycycline or tetracycline then erythromycin 500 mg po 4 times per day Pregnant: Desensitize and then treat with penicillin |
Late latent, tertiary or syphilis of unknown duration (except neurosyphilis) |
Adults: benzathine penicillin 2.4 million units x 3 administered one week apart for a total of 7.2 million units. Children: benzathine penicillin G 50,000 units/kg administered as three doses at one week intervals for a total of 150,000 units/kg, not to exceed 7.2 million units administered |
Nonpregnant; doxycycline 100 mg bid or tetracycline 500 mg 4 times per day x 28 days Pregnant: Desensitize and then treat with penicillin |
| Neurosyphilis | Adults: penicillin G 3-4 million units iv every 4 hours for a total of 18-24 million units per day or penicillin G procaine 2-4 million units im per day plus probenecid 500 mg po 4 times per day for 10-14 days. Alternative: Procaine penicillin G im 2.4 million units daily and probenecid 500 mg 4 times per day for 7-10 days |
Desensitization and then treat with penicillin. |
Table 4. Alternative Therapies for Primary, Secondary, and Latent Syphili
| DRUG AND ROUTE | DOSE | CSF LEVELS |
|---|---|---|
| Amoxicillin probenecid po | 3 gms bid (and probenecid 500 mg 4 times per day) for 7-10 days | Probably adequate |
| Ceftriaxone iv | 1 gm daily for 7-10 days | Adequate |
| Doxycycline po | 100 mg bid for 14-28 days | Probably adequate |
Figure 1. Chancre of Primary Syphilis
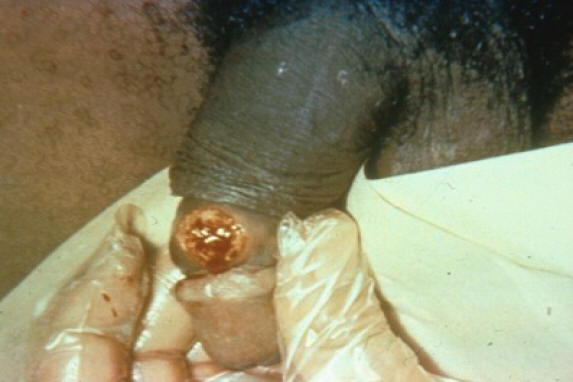
http://www.cdc.gov/std/training/clinicalslides/PowerPoint/syphilis.ppt#258,4,Primary syphilis-chancre
Figure 2. Rash of Secondary Syphilis

http://www.cdc.gov/std/training/clinicalslides/PowerPoint/syphilis.ppt#264,11,Secondary syphilis
What's New
Sena AC, et al. Response to Therapy Following Retreatment of Serofast Early Syphilis Patients with Benzathine Penicillin.; Clin Infect Dis. 2013;56:420-422.
Ghanem KG et al.Lumbar Puncture in HIV-infected Patients with Syphilis and No Neurologic Symptoms.Clin Infect Dis. 2009 Mar 15;48(6):816-21.
Harper KN, et al.On the origin of the trepanomatoses: a phylogenetic approach. PLOS Neglected Tropical Diseases 2008;2:e148
Hingwe, A Jarisch-Herxheimer reaction. 2009
Suspicious Skin Lesions and Secondary Syphilis.Infect Med. 2009:104-8.
Guided Medline Search For:
Reviews
Clement ME, Okeke L, Hicks, CB. Treatment of Syphilis: A Systematic Review. JAMA. 2014;312:1905-17.
Stamm LV. Global Challenge of Antibiotic-Resistant Treponema pallidum.; Antimicrobial Agents and Chemotherapy 2010;54:583-589.
Lynn WA, Lightman S. Syphilis and HIV: a dangerous combination. Lancet Infect Dis. 2004 Jul;4(7):456-66.
Guided Medline Search For Recent Reviews
History
Benedek TG: Biography of Neisser
Rothschild BM. History of Syphilis. Clin Infect Dis 2005;40:1454-1463.
Laura J. McGough. Demons, Nature or God? Witchcraft Accusations and the French Disease in Early Modern Medicine
Paul A. Lombardo and Gregory M. Dorr. Eugenics, Medical Education, and the Public Health Service: Another Perspective on the Tuskegee Syphilis Experiment
Francis AM. The wages of sin: how the discovery of penicillin reshaped modern sexuality. Arch Sex Behav 2013;42:5-13.
Guided Medline Search For Historical Aspects
Treponema pallidum (Syphilis)Table of Contents
- General Information
- Microbiology
- Epidemiology
- Clinical Manifestations
- Laboratory Diagnosis
- Direct Detect Methods
- Serological Tests
- Nontreponemal Reaginic Antibody Tests
- Fluorescent Treponemal Antibody-Absorption (FTA-abs)
- TPHA and MHATP
- Enzyme-linked Immunosorbent Assay (ELISA)
- Immunochromatographic Membrane Test (ICT)
- Immunochromatographic Strip (ICS)
- Line immunoassay (LIA)
- Testing Algorithms for Syphilis in Adults except Neurosyphilis
- Testing Algorithms for Congenital Syphilis
- Testing Algorithms for Neurosyphilis
- Pathogenesis
- Susceptibility In Vitro and In Vivo
- Antimicrobial Therapy
- Endpoint for Monitoring Therapy
- Vaccines
- Prevention
